Research Article - (2021) Volume 0, Issue 0
Cytotoxic effect and chromosomal damages induced by the methanolic extract of Cytisus triflorus assessed by Allium test
M. Nassar1,2*, R. Boulassel1, N. Bouhabel1, S. Djeghader1 and K. Seghir1Abstract
Cytisus triflorus is a medicinal plant that has been used for centuries in Algeria. The species' aerial part is commonly used to treat some abdominal diseases and wound healing. We assessed the cytotoxic/anticytotoxic and genotoxic/antigenotoxic activities of Cytisus triflorus (C.t) crude extract and their ability to protect meristematic cells from H2O2-induced nuclear damage. Three concentrations of the extract (0.25 mg/ml, 0.5 mg/ml and 1 mg/ml) were tested in the absence and the presence of H2O2 (2.5% and 5%). The test was applied to Allium sativum roots for 24 h. The extract was cytotoxic, especially at a 1 mg/ml concentration. A slowdown in root growth reflects this. The mitotic index was reduced up to 33% and 7% for the concentrations 0.5 mg/ml and 1 mg/ml (p<0.001), respectively, compared to the negative control (50%). However, an increase in the frequency of chromosomal aberrations was noted with the three concentrations of the extract (p<0.05 at 0.5 mg/ml). Furthermore, treating cells with the Extract-H2O2 mixture (at all concentrations) resulted in a decrease in the mitotic index as well as an increase in the frequency of chromosomal aberrations (p<0.05, stickiness, chromosomal bridge, and vagrant chromosome were the most detected aberrations in all the groups). These findings suggest that the extract has cytotoxic and genotoxic effects on Allium sativum cells, particularly at high concentrations.
Keywords
Cytisus triflorus, Hydrogen peroxide, Cytotoxic, Genotoxic, Mitotic index.
Introduction
Medicinal plants have been used for centuries as a remedy for various human diseases, and their therapeutic power is attributed to the active substances they contain (Tyihák et al., 2007). They continue to be the primary source of new drugs and are regarded as a critical source of raw material to discover new molecules required for drug development (Andriamparan et al., 2014). This assumes that plant-based products are not toxic, and therefore their use is done without safety and toxicology rules. As a result, several studies have shown that this assumption is incorrect, and using medicinal herbs can be harmful to human health.
Plant extracts used in traditional medicine may have genotoxic and cytotoxic effects that are toxic, mutagenic, and carcinogenic (Mengs, 1988; Ferreira-Machado et al., 2004). According to many authors (De Ferreira and Ferrro Vargas, 1999; AskinCelik and Aslanturk, 2007), long-term use of some herbs may result in potential mutagenic or genotoxic risks, and their use is associated with high rates of tumor formation in some persons. As a result, studies on herbal medicines should be conducted to evaluate their biological activities using appropriate biological and toxicological tests and chemical screening methods (Tyihák et al., 2007).
Due to its geographical location and bioclimatic diversity, Algeria has significant potential regarding aromatic and medicinal plants, which have been used for a long time in traditional medicine. Their use is inspired by the knowledge and the experience of local populations. However, this use does not follow any rules to ensure the safety and activity of these natural products. The Fabaceae family provides many drugs sought for various purposes (Chopra et al., 1960). The Fabaceae family has about 339 species grouped into 53 genera in Algeria. The genus Cytisus (Leguminosae) is widespread all across the Mediterranean, with 70 species, of which 8 are growing in northern Algeria. According to numerous research (Nirmal et al., 2008), this genus is used in traditional medicine for diuretic and hypertension treatment.
Cytisus triflorus L'Hérit, a member of the genus Cytisus, is known as "illougui" in Algeria. Cytisus triflorus is distributed throughout the Mediterranean region, and it is very abundant in the northeast of Algeria, especially in the mountains of Skikda (Quezel and Santa, 1963). The species is a branched shrub without thorns with a height of over 2 m. The stems and leaves are very silky, composed of 3 small leaves, oval, large in the middle, and can reach 3 cm length. The flowers, long of more than 1.5 cm, have pedicels, generally by groups of 3 to the axil of the superior leaves (Beniston, 1984). The aerial part of the species is commonly used to treat abdominal diseases and promote healing of wounds and injuries, the leaves are a deep green color used to dye sticks and hair. The bloom takes place from March till May.
Only a few investigations relate to this species's biological activities and toxicological effects. Therefore, the present study is the first to evaluate the cytotoxic and genotoxic potential of the crude extract obtained from the aerial parts of Cytisus triflorus using Allium sativum test.
Materials and Methods
Cytisus triflorus as plant sample
The aerial parts of Cytisus triflorus were collected in January 2021 from the region of Ainekechra, northwest of Skikda, the region is considered a virgin area where human activity is minimal, and its mountains are rich in medicinal plants. The aerial parts were dried at room temperature for 21 days before being ground into a fine powder with a blender.
Extraction
80 g of the aerial parts powder was put in the hydroalcoholic mixture (MeOH: H2O, 7:3 v/v). After 48 h, the obtained macerate was filtered and placed in a rotary vacuum evaporator to condense the hydroalcoholic extract. The crude extract was weighed and kept at 4°C until needed.
Cytotoxicity test
After obtaining a completely dry extract, three concentrations (0.25 mg/ml, 0.5 mg/ml and 1 mg/ml) were prepared from a stock solution of 4 mg/ml. Two concentrations of hydrogen peroxide (positive control) were prepared from a pure solution (30%). Initially, garlic cloves were selected, rinsed then placed in pillboxes containing distilled water to germinate. Cloves with root sizes ranging from 1to 1.5 cm were exposed to various concentrations of the extract alone (0.25 mg/ml, 0.5 mg/ml and 1mg/ml). The incubation lasted 3 days (72 h) in the dark with the renewal of the solution every 24 h. Root size is measured every 24 h.
Genotoxicity test
Cloves with a root length ranging from 1 to 1.5 cm were incubated with three concentrations (0.25 mg/ml, 0.5 mg/ml and 1mg/ml) of the extract with and without H2O2 (2.5% and 5%), the incubation takes place in the dark for 24 h.
Cytogenetic preparation
The roots of each treatment were recovered and fixed in a mixture (ethanol/acetic acid, 3v/1v) for 24 h before being transferred to 70% Ethanol and stored at 4°C until use. The roots are hydrolyzed in HCl 1N at 60°C for 10 min to prepare the slides (Bagatini et al., 2009; Soodan et al., 2017). After staining the roots with Schiff reagent for 20-30 min, the root tips were crushed between slides and coverslips in 45% acetic acid drop.
Parameters of genotoxicity
Normal and abnormal cells were counted from at least 10 root tips collected from several cloves (n=5, for each treatment), with over 600 cells counted in each root tip. The cells were examined using a Primostar microscope (Zeiss) at magnification X400.
Mitotic index

Index of chromosomal aberration
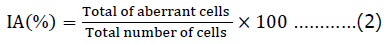
Statistical analysis
ANOVA test was used for all statistical analyses. The posthoc Tukey test was used to examine the difference in group averages using SPSS version 14.0 software. The results were reported as Mean and Standard Deviation, with significant differences at p<0.05.
Results
The results indicated in Table 1 revealed that the root growth of garlic cloves treated with 0.25 mg/ml and 0.5 mg/ml continues during the 3 days (72 h) of treatment. However, the growth rate slowed down; the root sizes were 2.35 cm and 1.99 cm, respectively, compared to the control (3.31 cm). The treatment of cloves with the highest concentration 1 mg/ml led to a considerable decrease in root growth (1.5 cm) compared to control. The root length is significantly shorter than the control (P<0.05). This implies that this concentration is both inhibitory and cytotoxic. As shown in Table 1, the inhibition of root elongation was increased by increasing the concentration of the extract. In terms of shape, the roots appeared softer and more fragile, with a brownish color, in particular at the concentration 0.5 mg/ml and 1 mg/ml.
| Treatment and concentrations | Root length (0 h) | Root length (72 h) | Root growing (72 h) | Growing (%) | Inhibition (%) | IC50 (mg/ml) |
|---|---|---|---|---|---|---|
| Control | 1.2 ± 0.3 | 3.31 ± 0.45 | 1.91 | 100 | 0 | - |
| H2O2 2.5% | 1.1 ± 0.24 | 2.26 ± 0.4 | 1.16 | 61.90 | 38.9 | |
| H2O2 5% | 1.0 ± 0.29 | 2.05 ± 0.49 | 1.05 | 50 | 50 | |
| C. t 0.25 mg/ml | 1.11 ± 0.27 | 2.35 ± 0.47 | 1.24 | 65.26 | 11.91 | 0.64 |
| C. t 0.5 mg/ml | 1.07 ± 0.38 | 1.99 ± 0.4 | 0,92 | 48.42 | 47.62 | |
| C. t 1 mg/ml | 1.0 ± 0.43 | 1.5 ± 0.35* | 0.5 | 26.31 | 76.2 |
Table 1. Root growth of Allium sativum after three days (72 h) of treatment with the three concentrations of Cytisus triflorus extract.
As illustrated in Table 2, the rate of cell division varies depending on the concentrations tested and the treatments. At 0.25 mg/ml of the extract, the rate of mitotic division is critical (49%), and almost identical to control (51%). The mitotic index (MI) decreases up to 28% at the concentration of 0.5 mg/ml of the extract. However, when compared to control, the highest concentration, 1 mg/ml has a highly significant inhibitory effect (p<0.001) on the rate of mitotic division (4.98%, Table 2). The inhibitory effect of the concentrations, 0.5 mg/ml and 1 mg/ml on the mitotic division was higher than that of hydrogen peroxide (H2O2) at the concentrations 2.5% and 5%, in which the mitotic index was respectively, 51% and 38%. In addition, the rate of the mitotic division was lowered in the roots of Allium sativum treated with various concentrations of the extract (0.25 mg/ml, 0.5 mg/ml, and 1 mg/ml) combined with H2O2 (2.5% and 5%) but not considerably, MI>30% in all groups.
| Treatments | MI | Cells in Division (%) | ||||
|---|---|---|---|---|---|---|
| Interphase | Prophase | Metaphase | Anaphase | Telophase | ||
| Control | 51.77 ± 4.6 | 48.43 ± 6.4 | 16.1 ± 4.9 | 18.27 ± 3.3 | 6.07 ± 3.28 | 11.4 ± 4.1 |
| C.t 0.25 mg/ml | 49.03 ± 5.7 | 51.01 ± 9.8 | 3.7 ± 2.9 | 22.3 ± 7.7 | 4.4 ± 5.5 | 18.63 ± 3.2 |
| C.t 0.5 mg/ml | 28 | 71.1 ± 9.2 | 2.4 ± 4.2* | 13.5 ± 4.0 | 4.4 ± 3.5 | 7.8 ± 3.9 |
| C.t 1mg/ml | 4.98 ± 2.8*** | 95.02 ± 8.9 | 0.63 ± 1.3*** | 2.76 ± 3.3*** | 0.96 ± 1.2*** | 0.63 ± 0.9*** |
| 2.5% H2O2 | 51.4 ± 4.5 | 48.6 ± 8.2 | 11.7 ± 5.4 | 25.1 ± 3.7 | 8.08 ± 3.28 | 6.7 ± 3.8 |
| 5% H2O2 | 38.43 ± 6.7 | 71.57 ± 5.7 | 3.3 ± 3.9 | 16.2 ± 8.6 | 3.3 ± 1.72 | 5.63 ± 2.19 |
| C.t 0.25 mg/ml+2.5% H2O2 | 40.85 ± 9.6 | 59.15 ± 10.9 | 5.96 ± 3.7 | 18.8 ± 8.2 | 7.02 ± 4.0 | 9.07 ± 4.3 |
| C.t 0.25 mg/ml+5% H2O2 | 31.6 ± 11.1 | 71.29 ± 23.2 | 3.27 ± 3.6 | 11.06 ± 12.9 | 6.95 ± 2.6 | 10.3 ± 2.2 |
| C.t 0.5 mg/ml+2.5% H2O2 | 31.9 ± 3.4 | 68.1 ± 5.4 | 2.86 ± 4.1 | 1 ± 1.4 | 2.3 ± 1.2 | 10.76 ± 4.95 |
| C.t 0.5 mg/ml+5% H2O2 | 34.4 ± 9.2 | 65.6 ± 8.9 | 2.56 ± 3.7 | 18.5 ± 10.4 | 2.6 ± 1.4 | 10.7 ± 5.6 |
| C.t 1 mg/ml+2.5% H2O2 | 30.02 ± 8.2 | 69.98 ± 11.4 | 1.8 ± 4.3 | 16.6 ± 12.6 | 1.2 ± 1.6 | 10.4 ± 7.1 |
| C.t 1 mg/ml+5% H2O2 | 30.3 ± 8.8 | 69.7 ± 16.9 | 2.93 ± 5.3 | 13.16 ± 9.6 | 4.33 ± 2.9 | 10.23 ± 6.4 |
Table 2. The effect of Cytisus triflorus extract alone and in combination with H2O2 on Allium sativum mitotic division. ***p<0.001, the treated groups compared to control, C.t: Cytisus triflorus.
Regarding the frequency of the different division phases, the data revealed that the extract reduced the percentage of cells in prophase and considerably enhanced the percentage of cells in metaphase. These findings imply that the methanolic extract of Cytisus triflorus involves antimitotic activity while blocking cells in metaphase (Table 2).
The results indicate that both concentrations of 0.5 mg/ml and 1 mg/ml of Cytisus triflorus extract inhibit the cell division of Allium sativum. This is expressed by slower root growth when compared to the negative control.
Cytogenetic analysis of Allium sativum root tips revealed an increase in the chromosomal aberration index in all the groups. In cells treated with Cytisus triflorus extract alone, the chromosomal aberration rate increases proportionally to the concentration, particularly at 0.5 mg/ml, the chromosomal aberration index value reported as 6.24% (P<0.05), which is very close to that of the positive control (H2O2) at both concentrations 2.5% and 5% (Table 3). The highest concentration (1 mg/ml) causes a significant decrease in the chromosomal aberration index, which is closely related to a decrease in the rate of mitotic division.
| Groups | St | Fg | Mn | B | Mp | Cm | V | Bn | AI (%) |
|---|---|---|---|---|---|---|---|---|---|
| Control | 0 | 0 | 0.03 | 0.13 ± 0.59 | 0 ± 0 | 0.3 ± 0.59 | 0.13 ± 0.4 | 0 ± 0 | 0.56 ± 1.8 |
| H2O2 2.5% | 3.0 ± 1.4* | 0.13 ± 0.14 | 0 | 0.33 ± 0.91 | 0.5 ± 1.62 | 0.24 ± 2.1 | 3.3 ± 3.1* | 0 ± 0 | 7.46 ± 3.3* |
| H2O2 5% | 4.08 ± 1.9* | 0.13 ± 0.8 | 0.03 ± 0.4 | 1.24 ± 1.30 | 0.20 ± 0.17 | 0.96 ± 0.87 | 0.96 ± 0.27 | 0 ± 0 | 7.65 ± 2.26* |
| C.t 0.25 mg/ml | 2.2 ±1.6 | 0 ± 0 | 0 ± 0 | 0.53 ± 2.0 | 0.43 ± 0.5 | 0.33 ± 0.7 | 0.63 ± 0.63 | 0 ± 0 | 4.12 ± 1.76 |
| C.t 0.5 mg/ml | 3.53 ± 0.9* | 0.06 ± 0.12 | 0.16 ± 0.2 | 0.44 ± 0.97 | 0.33 ± 0.2 | 0.46 ± 0.2 | 1.17 ± 1.45 | 0 ± 0 | 6.24 ± 4.5* |
| C.t 1 mg/ml | 0.93 ± 0.76 | 0 ± 0 | 0.06 ± 0.06 | 0.03 ± 0 | 0.03 ± 0.05 | 0.06 ± 0.05 | 0.1 ± 0.73 | 0.16 ± 0.2 | 1.37 ± 1.3 |
| C.t 0.25 mg/ml+H2O2 2.5% | 5.83 ± 2.21* | 0.07 ± 0.09 | 0.37 ± 0.63 | 3.13 ± 0.1* | 0.23 ± 0.16 | 0.57 ± 0.97 | 3.33 ± 1.76* | 0 ± 0 | 13.5 ± 3.1* |
| C.t 0.5 mg/ml+H2O2 2.5% | 9.5 ± 2.43* | 0.17 ± 0.73 | 0.10 ± 0.1 | 0.93 ± 1.6 | 0.23 ± 0.21 | 0.47 ± 0.84 | 3.90 ± 2.61* | 0 ± 0 | 15.3 ± 4.2* |
| C.t 1 mg/ml+H2O2 2.5% | 9.87 ± 4.87* | 0.03 ± 0.05 | 0 ± 0 | 0.30 ± 0.9 | 0.10 ± 0.5 | 0.07 ± 0.1 | 0.80 ± 0.3 | 0 ± 0 | 11.2 ± 2.6* |
| C.t 0.25 mg/ml+H2O2 0.5% | 6.73 ± 3.26* | 0.07 ± 0.05 | 0.20 ± 0.8 | 2,47 ± 0.23* | 0.13 ± 0.3 | 0.5 ± 0.3 | 0.83 ± 1.12* | 0 ± 0 | 14 ± 4.3* |
| C.t 0.5 mg/ml+H2O2 0.5% | 21.47 ± 1.5*** | 0.03 ± 0.15 | 0.07 ± 0.27 | 1.0 ± 2.3 | 0.87 ± 1.3 | 0.27 ± 0.3 | 3.37 ± 1.3* | 0.20 ± 0.6 | 27.2 ± 5.3*** |
| C.t 1 mg/ml+H2O2 0.5% | 9.26 ± 0.43* | 0.07 ± 0.27 | 0.07 ± 0.27 | 0.93 ± 0.6 | 1.2 ± 1.5 | 0.53 ± 0.77 | 3.77 ± 2.15* | 0 ± 0 | 12.8 ± 3.1* |
Table 3. The frequency of various types of chromosomal aberrations detected in Allium sativum cells.
Furthermore, all cells exposed to H2O2 in the presence or absence of Cytisus triflorus extract showed a significant increase in the chromosomal aberration index (Table 3), especially at 5% of H2O2 combined with 0.5 mg/ml of the extract (IA=27%, P<0,001). Besides, incubating cells to different concentrations of the Extract-H2O2 mixture did not repair or decrease the rate of nuclear damage caused by H2O2. On the contrary, all concentrations significantly increased the total number of chromosomal aberrations compared to a positive control (H2O2). Based on these findings, it seems that the methanolic extract of Cytisus triflorus, exerts a genotoxic effect and that of H2O2, leading to the appearance of chromosomal aberrations and increasing their frequency as well.
In our study, different types of chromosomal aberration (Sticky chromosomes, vagrant chromosomes, chromosomal bridge, micronuclei, and C-mitosis) detected Allium sativum cells by cytogenetic analysis. However, their frequency depends on the concentrations and treatments.
In general, the treatment of cells with the extract alone increased the frequency of each chromosomal aberration compared to the negative control; this increase was positively correlated with the concentration. At the highest concentration of the extract (1 mg/ml), the increase was minor as the rate of mitotic division reduced considerably at this concentration (p<0,001).
The treatment of cells with the extract at a concentration of 0.5 mg/ml resulted in a significant increase in the total number of aberrant cells as well as the type of sticky chromosomes (p<0.05) (Table 2). Cells treated with H2O2 and Cytisus villosus extract at all concentrations had the highest frequency of chromosomal aberrations (Fig. 1).
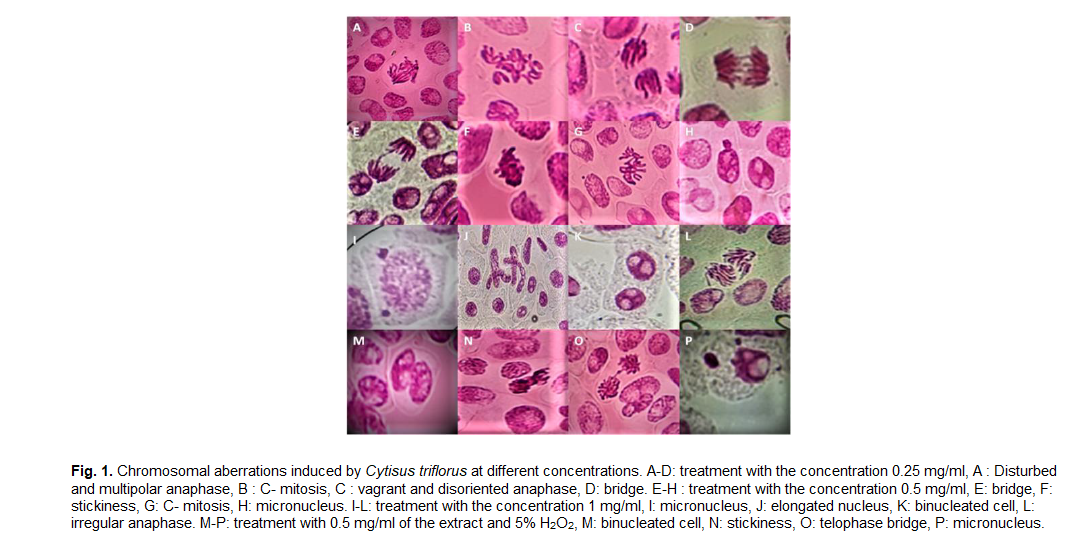
Fig 1. Chromosomal aberrations induced by Cytisus triflorus at different concentrations. A-D: treatment with the concentration 0.25 mg/ml, A: Disturbed and multipolar anaphase, B: C- mitosis, C: vagrant and disoriented anaphase, D: bridge. E-H: treatment with the concentration 0.5 mg/ml, E: bridge, F: stickiness, G: C- mitosis, H: micronucleus. I-L: treatment with the concentration 1 mg/ml, I: micronucleus, J: elongated nucleus, K: binucleated cell, L: irregular anaphase. M-P: treatment with 0.5 mg/ml of the extract and 5% H2O2, M: binucleated cell, N: stickiness, O: telophase bridge, P: micronucleus.
The frequency of sticky chromosomes was significant at all concentrations, while the frequency of chromosomal bridges and vagrant chromosomes was significant at most concentrations (Table 3).
Discussion
The purpose of this study was to assess the cytotoxicity and the genotoxicity of Cytisus triflorus extract collected from the western coastal region of Skikda. According to the cytotoxicity results, the methanolic extract of Cytisus triflorus is slightly cytotoxic at low concentrations (0.25 mg/ml and 0.5 mg/ml), whereas a higher concentration (1 mg/ml) is substantially cytotoxic (P<0.05). All concentrations inhibited root growth, especially during the two last days of incubation. According to Egito et al. (2007), root elongation inhibition is regarded as a sign of toxicity, commonly associated with a decrease in cell division. Root growth inhibition exerted by the extract may be related to an allelochemical action of the polyphenols in the extract. According to Li et al. (2011), phenolic compounds can influence nutrient uptake by roots, resulting in inhibition of root elongation and development.
Cytisus triflorus methanolic extract exerts an antimitotic action by preventing many cells from entering prophase or blocking metaphase cells. A reduction in cell mitotic activity accompanied the inhibition of root growth. Among all groups in the experiment, cells treated with both concentrations of 0.5 mg/ml and 1 mg/ml exhibited the lowest mitotic index (P<0.001). In addition, the mitotic index value decreased when cells were treated with the extract/H2O2 combination, which was confirmed at all concentrations tested. Many plant extracts have previously been shown to decrease mitotic index and DNA and protein synthesis (Schulze and Kirschner, 1986; Celik and Aslanturk, 2010; Tajudeen et al., 2020).
The mitotic index is a reliable indicator of cytotoxicity in all living organisms. The results revealed that a decrease in root length is associated with a decrease in mitotic activity; this could be due to inhibition of DNA synthesis or a blockage in the G2 phase of the cell cycle, preventing the cell from entering mitosis (Askn and Aslanturk, 2007). This could also be due to altered nucleoprotein synthesis and a lack of ATP, preventing spindle elongation, microtubule dynamics, and chromosomal movement (Lazareva et al., 2003). The study conducted by (Boughrara, 2016) showed that the crude methanolic extract of the aerial part of Cytisus triflorus, is rich in alkaloids, anthocyanins, cardenolides, flavonoids, quinones, tannins, saponins. It has been shown that some alkaloids can act as herbicides by inhibiting the cell proliferation of competing plants and consequently inhibiting their growth (Vanderplanck and Glauser, 2018). These inhibitory effects may be related to the biochemical composition of the secondary molecules found in the extract.
According to (Muthulakshmi et al., 2014), H2O2 can be toxic to cells, it is tightly related to oxidative stress due to reactive oxygen species exposure (ROS). These molecules can easily adhere to DNA molecules, blocking cell division and damaging DNA by oxidation.
These results are consistent with those obtained by (Djidel et al., 2018), showing the toxicity of the aqueous extract of Cytisus triflorus towards the adults of Tribolium castaneum species at different concentrations (5%, 10%, 20%). The toxicity of some species belonging to the genus Cytisus has already been reported, namely the species Cytisus scoparius and Cytisus canariensis, due to their richness in alkaloids, especially (cytisine and sparteine) (Wink, 2009). According to Wink (2000), many secondary metabolites, including alkaloids, can attack nucleic acids (DNA and RNA), either by intercalation or alkylation. These compounds stabilize DNA and thus inhibit its replication. If the alkylated DNA bases are not repaired, they can cause mutations and even cancer (Wink, 2009).
The results revealed that Cytisus triflorus extract is genotoxic at the concentrations tested in chromosomal aerations. The percentages of aberrations observed following treatment with the extract are significantly higher than the negative control, especially at the concentration of 0.5 mg/ml (AI=6.24%, p<0.05). Furthermore, the extract did not induce any antigenotoxic effect towards the genotoxicity of hydrogen peroxide. The rates of chromosomal aberrations observed during the treatment of the cells with the combination of hydrogen peroxide/extract were even higher than those of the positive control, which indicates that the extract amplified the genotoxic effect of hydrogen peroxide, especially at the concentration (5% H2O2 and 0.5 mg/ml extract together). Hence, the chromosomal aberration index was 27.2%, 3.5 times higher than that of the positive control's value of 5% (7.65%).
Stickiness, vagrant chromosome, and bridge are the most common chromosomal abnormalities in our study. These chromosomal aberrations are caused by chromosomal material rupture or exchange and result in chromosomal location and structure changes. The majority of these aberrations are fatal. However, numerous related mutations are still viable and can have negative genetic, somatic, or hereditary effects (Swierenga et al., 1991). Stickiness, vagrant chromosome, and C-mitosis are all induced by aneugenic effects (Fiskesjö, 1985; Singh, 2003). Stickiness is described as chromosome agglomeration caused by DNA degradation or depolymerization, resulting in a sticky aspect of chromosomes (Souguir et al., 2008; Yildiz and Arikan, 2008). Even though the chromosomal bridge is a clear example of clastogenicity and genotoxicity, they are formed between chromosomes or isolated chromosome fragments caused by single or double-stranded breaks (Singh, 2003). In contrast, the induction of vagrant chromosomes results in separating unequal numbers of chromosomes in daughter cells with unequal size and shape nuclei during interphase (Singh, 2003). C-mitosis is also a result of microtubule disruption inducing mitotic spindle inactivation (Liman et al., 2010). Based on the data collected, it is quite clear that Cytisus triflorus extract (0.25 mg/ml, 0.5 and 1 mg/ml) disrupts the cell cycle and affects chromatin organization and DNA replication resulting in the appearance of various types of chromosomal abnormalities. The increase in chromosomal abnormalities after incubation of cells with the Extract-H2O2 mixture, is reflected by the extract's inability to regulate and destroy reactive oxygen species produced by H2O2. All these data suggest that the extract of Cytisus triflorus is cytotoxic and genotoxic, besides the concentration of 0.25 mg/ml, where the effects are slight and statistically insignificant.
Conclusion
According to the findings of this investigation, the methanolic extract of Cytisus triflorus, commonly used in traditional medicine, clearly demonstrates cytotoxic and genotoxic effects, especially at high concentrations (0.5 mg/ml and 1 mg/ml). These effects are manifested as root growth inhibition, a decrease in mitotic index, induction of chromosomal aberrations. As a result, precautions must be taken when using this extract as a traditional remedy. However, additional cytogenetic in vitro and in vivo experiments towards the cytotoxicity and genotoxicity of this extract using other test models (animals, cell lines) are needed to establish procedures for the biological use of this extract.
Acknowledgment
The authors are grateful to the MESRS, Algeria.
References
Andriamparan, J.N., Brinkmann, K., Jeannoda, V., Buerkert, A. (2014). Effects of socio-economic household characteristics on traditional knowledge and usage of wild yams and medicinal plants in the Mahafaly region of south-western Madagascar. Journal of Ethnobiology and Ethnomedicine, 10:61-69.
AskinCelik, T., Aslanturk, O.S. (2007). Cytotoxic and genotoxic effects of Lavandula stoechas aqueous extracts. Biologia, 62:292-296.
Bagatini, M.D., Fachinetto, J.M., Ferreira da Silva, A.C., Tedesco, S.B. (2009). Cytotoxic effects of infusions (tea) of Solidagomicro glossa DC. (Asteraceae) on the cell cycle of Allium cepa. Revista Brasileira de Farmacognosia Rev bras Farmacogn, 19:632-636.
Beniston, N.t. (1984). Fleurs d’algerie. Entrepise nationale du livre, Alger.
Boughrara, B. (2016). Inventaire et etude ethnobotanique et chimique des plantes à interest thérapeutique et nutritif du parc national El-Kala. PhD Thesis, p:53.
Celik, T.A., Aslanturk, O.S. (2010). Evaluation of cytotoxicity and genotoxicity of inula viscosa leaf extracts with Allium test. Journal of Biomedicine and Biotechnology, 8:1-8.
Chopra, C., Abrol, B.K.K., Handa, L. (1960). Les plantes médicinales des regions arides considérées surtout du point de vue botanique. UNESCO, Paris.
Djidel, A., Daghbouche, S., Benrima, A., Djazouli, Z.E. (2018). Evaluation de l’activité insecticide de l’extrait aqueux brut de la fabacae Cytisus triflorus L’HER á l’égard de Tribolium castanium (herbst, 1797) (Coleoptera: tenebrionidae). Revue Agrobiologia, 8:1093-1110.
Egito, L.C.M., Medeiros, M.G., Medeiros, S.R.B., Agnezlima, L.F. (2007). Cytotoxic and genotoxic potential of surface water from the Pitimbu river, northeastern/RN Brazil. Genetics and Molecular Biology, 30:435-441.
Fernandes, De.S., Ferreira, I.C., Ferraro Vargas, V.M. (1999). Mutagenicity of medicinal plant extracts in Salmonella/microsome assay. Phytotherapy Research, 13:397-400.
Ferreira-Machado, S.C., Rodrigues, M.P., Nunes, A.P.M. (2004). Genotoxic potentiality of aqueous extract prepared from Chrysobalanus icaco L. Leaves. Toxicology Letters, 151:481-487.
Fiskesjö, G. (1985). The Allium test as a standard in environmental monitoring. Hereditas, 102:99-112.
Lazareva, E.M., Polyakov, V.Y., Chentsov, Y.S., Smirnova, E.A. (2003). Time and cell cycle dependent formation of heterogeneous tubulin arrays induced by colchicines in Triticum aestivum root meristem cell. Biology International, 27:633-646.
Li, Z.H., Wang, Q., Ruan, X., Pan, C.D., Jiang, D.A (2010). Phenolics and plant allelopathy. Molecules, 15:8933-8952.
Liman. R., Akyil, D., Eren, Y., Konuk, M. (2010). Testing of the mutagenicity and genotoxicity of metolcarb by using both Ames/Salmonella and Allium test. Chemosphere, 80:1056-1061.
Mengs, U. (1988). Toxic effects of sennosides in laboratory animals and in vitro. Pharmacology, 36:180-187.
Muthu Lakshmi, L.T., Radha, R., Jayshree, N. (2014). In vitro antioxidant activity. Total phenolic and total flavonoid content in extracts from the bark of Dalbergiasissoo Roxb. International Journal of Pharmaceutical Sciences and Research, 5:226-231.
Nirmal. J., Babu, C.S., Harisudhan, T., Ramanathan, M. (2008). Evaluation of behavioural and antioxidant activity of Cytisus scoparius Link in rats exposed to chronic unpredictable mild stress, BMC Complement. Journal of Alternative and Complementary Medicine, 8:1-8.
Quezel, P., Santa, S. (1963). Nouvelle flore de l’Algérie et des régions désertiques meridionales. CNRS, Paris.
Schulze, E.S., Kirschner, M.W. (1986). Microtubule dynamics in interphase cells. Journal of Cell Biology, 102:1020-1031.
Singh, R.J. (2003). Plant Cytogenetics. CRC Press, Boca Raton, Florida.
Soodan, R.K., Sharma, A., Kaur, M., Katnoria, J.K, Nagpal, A.K (2017). Allium cepa root chromosomal aberration assay an application in assessing antigenotoxic potential of ashwagandha in: Science of Ashwagandha. Preventive and Therapeutic Potentials, pp:69-79.
Souguir, D., Ferjani, E., Ledoigt, G., Goupil, P. (2008). Exposure of Vicia faba and Pisum sativum to copper induced genotoxicity. Protoplasma, 233:203-207.
Swierenga, S.H.H., Heddle, J.A., Sigal, E.A. (1991). Recommended protocols based on a survey of current practice in genotoxicity testing laboratories IV. Chromosome aberration and sister chromatid exchange in Chinese hamster ovary. V79 Chinese hamster lung and human lymphocyte cultures. Mutation Research, 246:301-322.
Tajudeen, A., Owolarafea, K., Salawua, G.O., Ihegboroa, C.J., Ononamadua, A.J., Alhassanb, A.M., Wudilb, A.M. (2020). Investigation of cytotoxicity potential of different extracts of Ziziphus mauritiana (Lam) leaf in Allium cepa model, Toxicology Reports, 7:816-821.
Tyihák, E., Móricz, Á.M., Ott, P.G. (2007). Biodetection and determination of biological activity of natural compounds. In Thin Layer Chromatography in Phytochemistry. CRC Press, pp:193-213.
Vanderplanck, M., Glauser, G. (2018). Integration of non-targeted metabolomics and automated determination of elemental compositions for comprehensive alkaloid profiling in plants. Phytochemistry, 154:1-9.
Wink, M. (2000). Interference of alkaloids with neuroreceptors and ion channels. Bioactive Natural Products, 11:123-129.
Wink, M. (2009). Mode of action and toxicology of plant toxins and poisonous plants. Mitt Julius Kühn-Inst, 421:93-112.
Yildiz, M., Arikan, E.V. (2008). Genotoxicity testing of quizalofopP-ethyl herbicide using the Allium cepa anaphase-telophase chromosome aberration assay. Caryologia, 61:45-52.
Author Info
M. Nassar1,2*, R. Boulassel1, N. Bouhabel1, S. Djeghader1 and K. Seghir12Laboratory of Research in Biodiversity Interaction, Ecosystem and Biotechnology' LRIBEB', University of 20 August 1955 BP 26, Route d'El Hadaiek-Skikda 21000, Algeria
Citation: Nassar, M., Boulassel, R., Bouhabel, N., Djeghader, S., Seghir, K. (2021). Cytotoxic effect and chromosomal damages induced by the methanolic extract of Cytisus triflorus assessed by Allium test. Ukrainian Journal of Ecology. 11:28-34.
Received: 05-Nov-2021 Accepted: 29-Dec-2021 Published: 05-Jan-2022
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
