Research Article - (2022) Volume 12, Issue 3
Five new species of teloschistaceae lichens from Algeria
R. Mouedden1*, A. Ayache1, A.H. Benchohra2, F. Toumi2 and F.Z. Bendimred1Abstract
Aim of this work is to describe the diversity of teloschistaceae in the forest of Doui Thabet (Saida province, western Algeria). The order Teloschistales contains species of nitrophilous lichens that grow rapidly on nitrogen-rich substrates. There are Teloschistaceae containing anthraquinones, Teloschistaceae with sedifolia grey and Physciaceae rich in atranorine. After sampling and collecting various specimens between 2019 and 2021 in this forest; the identification revealed 22 species of the Teloschistaceae family reported for the first time from this region. The following five species are noted: Blastenia xerothermica, Caloplaca intern, Pyrenodesmia microstepposa, Usnochroma carphineum, and Variospora glomerata are new species for Algeria.
https://www.mobafire.com/profile/ssplusyachting12-1122288?profilepage http://www.effecthub.com/user/3727843 http://hawkee.com/profile/5024697/ https://www.youmagine.com/ssplusyachting12/designs https://www.myminifactory.com/users/Ssplusyachting12 http://qooh.me/Ssplusyachting https://linktr.ee/ssplusyachting12 https://pubhtml5.com/homepage/spfvq/ https://telegra.ph/Ssplusyachting12-09-18 https://www.diggerslist.com/65085a6c796ab/about https://allmyfaves.com/Ssplusyachting12 https://www.metal-archives.com/users/Ssplusyachting12 https://www.catchafire.org/profiles/2501481/ https://www.fimfiction.net/user/638001/Ssplusyachting12 https://www.hebergementweb.org/members/ssplusyachting12.555456/ https://www.sqlservercentral.com/forums/user/ssplusyachting12 https://www.twitch.tv/ssplusyachting12/about https://www.roleplaygateway.com/member/Ssplusyachting12/ https://www.provenexpert.com/ssplusyachting12/?mode=preview https://www.intensedebate.com/people/Ssplusyachting https://www.indiegogo.com/individuals/35275554 https://visual.ly/users/ssplusyachting/portfolio https://slides.com/ssplusyachting12 https://letterboxd.com/Ssplusyachting1/ https://fliphtml5.com/dashboard/public-profile/zxpmg https://community.windy.com/user/ssplusyachting12 https://speakerdeck.com/ssplusyachting12 https://trello.com/u/ssplusyachting https://myanimelist.net/profile/Ssplusyachting12 https://3dwarehouse.sketchup.com/user/30ca4efb-4c09-4caf-ac85-7f103189253e/Ssplusyachting12-S https://www.wattpad.com/user/Ssplusyachting12 https://www.goodreads.com/user/show/170026239-ssplusyachting12 https://tr.gravatar.com/ssplusyachting https://tr.pinterest.com/ssplusyachting/ https://www.mapleprimes.com/users/Ssplusyachting12 https://medium.com/@ssplusyachting https://www.ted.com/profiles/45080180 https://www.infragistics.com/community/members/94d70ed75bb7249fda82b3be9b202d35647dca9d?_ga=2.196832940.1881464809.1695126162-1260905931.1695126162 https://www.metooo.io/u/65099dfd63f25c796f95ddb6 https://app.roll20.net/users/12437619/ssplusyachting12-s https://list.ly/ssplusyachting/activity https://giphy.com/channel/Ssplusyachting12 https://www.tumblr.com/ssplusyachting12 https://www.creativelive.com/student/ssplusyachting12-ssplusyachting12?via=accounts-freeform_2 https://gab.com/Ssplusyachting12 https://sketchfab.com/Ssplusyachting12 https://www.flickr.com/people/199148519@N05/ https://profiles.wordpress.org/ssplusyachting12/ https://wefunder.com/ssplusyachting12 https://seedandspark.com/user/ssplusyachting12-01hapvhx1447xvj1p12hqjz52g https://calis.delfi.lv/blogs/lietotajs/315023-ssplusyachting12/ https://os.mbed.com/users/ssplusyachting12/ https://notionpress.com/author/926905 https://my.desktopnexus.com/Ssplusyachting12/ https://guides.co/a/ssplusyachting12-ssplusyachting12/ https://www.bahamaslocal.com/userprofile/1/243967/Ssplusyachting12.html http://molbiol.ru/forums/index.php?showuser=1298935 https://www.credly.com/users/ssplusyachting12-ssplusyachting12/badges https://www.facer.io/user/icVEnlO5mr https://ko-fi.com/ssplusyachting12#paypalModal https://mastodon.online/@Ssplusyachting12 https://devpost.com/ssplusyachting?ref_content=user-portfolio&ref_feature=portfolio&ref_medium=global-nav https://www.redbubble.com/people/Ssplusyachting/shop?asc=u https://dzone.com/users/4995447/ssplusyachting12.html https://hub.docker.com/u/ssplusyachting12 http://onlineboxing.net/jforum/user/editDone/251907.page https://micro.blog/Ssplusyachting12 https://www.mobafire.com/profile/propertymarmaris12-1119729?profilepage https://www.wishlistr.com/profile/ http://www.effecthub.com/user/3724882 http://hawkee.com/profile/4910958/ https://www.youmagine.com/propertymarmaris12/designs https://www.myminifactory.com/users/propertymarmaris12 http://onlineboxing.net/jforum/user/edit/249190.page http://qooh.me/account/home/ https://band.us/band/92204743 https://linktr.ee/propertymarmaris12 https://pubhtml5.com/homepage/vrszv/ https://telegra.ph/propertymarmaris12-08-29 https://www.diggerslist.com/propertymarmaris12/about https://www.facer.io/user/gYpnOVrMDg https://allmyfaves.com/propertymarmaris12 https://www.metal-archives.com/users/propertymarmaris12 https://www.catchafire.org/profiles/2483499/ https://www.fimfiction.net/user/631649/propertymarmaris12 https://www.hebergementweb.org/members/propertymarmaris12.544810/ https://www.sqlservercentral.com/forums/user/propertymarmaris12 https://www.twitch.tv/propertymarmaris12/about https://www.roleplaygateway.com/member/propertymarmaris12/ https://ko-fi.com/propertymarmaris12 https://www.provenexpert.com/propertymarmaris12/ https://www.intensedebate.com/people/Propertymmm https://www.indiegogo.com/individuals/35086695 https://visual.ly/users/propertymarmaris/portfolio https://slides.com/propertymarmaris12 https://letterboxd.com/propertym/ https://micro.blog/propertymarmaris12 https://fliphtml5.com/dashboard/public-profile/bjmna https://community.windy.com/user/propertymarmaris https://www.longisland.com/profile/propertymarmaris12 https://speakerdeck.com/propertymarmaris12 https://trello.com/u/propertymarmaris/activity https://www.redbubble.com/people/propertym/shop?asc=u https://disqus.com/by/propertymarmaris12/about/ https://myanimelist.net/profile/propertymarmaris https://3dwarehouse.sketchup.com/user/cb673828-b6cf-4a0e-8208-d3d292972c53/propertymarmaris12-P?tab=models https://www.wattpad.com/user/propertymarmaris12 https://www.goodreads.com/user/show/169429527-propertymarmaris12 https://sourceforge.net/u/propertymarmari/profile https://en.gravatar.com/propertymarmaris0855255d4a https://tr.pinterest.com/propertymarmaris/ https://www.mapleprimes.com/users/Propertymarmaris12 https://medium.com/@propertymarmaris/about https://www.ted.com/profiles/44897843 https://www.infragistics.com/community/members/09de800e999893ac4d5c9789335b47c5b4a08fe5?_ga=2.47235623.1089752791.1693392418-317991250.1693392418 https://propertymarmari.livejournal.com/profile https://www.metooo.io/u/64ef20a26d5e6b4d2dc287ab https://hubpages.com/@propertymarmaris12 https://app.roll20.net/users/12357708/propertymarmaris12-p https://list.ly/propertymarmaris/ https://giphy.com/channel/Propertymarmaris12 https://www.tumblr.com/blog/propertymarmaris12 https://dzone.com/users/4985787/propertymarmaris12.html https://www.creativelive.com/student/propertymarmaris12-propertymarmaris12?via=accounts-freeform_2 https://gab.com/Propertymarmaris12 https://sketchfab.com/propertymarmaris12 https://www.flickr.com/people/199068945@N06/ https://devpost.com/propertymarmaris https://hub.docker.com/u/propertymarmaris12 https://forums.autodesk.com/t5/user/viewprofilepage/user/self https://profiles.wordpress.org/propertymarmaris12/ https://mastodon.online/@Propertymarmaris12 https://wefunder.com/delalabdullatifabzak https://seedandspark.com/user/propertymarmaris12 https://calis.delfi.lv/profils/lietotajs/312043-propertymarmaris12/ https://os.mbed.com/users/propertymarmaris12/ https://notionpress.com/author/919471 https://my.desktopnexus.com/propertymarmaris12/ https://guides.co/a/propertymarmaris12-propertymarmaris12/ https://www.bahamaslocal.com/userprofile/1/237941/propertymarmaris12.html http://molbiol.ru/forums/index.php?showuser=1295714 https://www.credly.com/users/propertymarmaris12-propertymarmaris12/badges https://orcid.org/0009-0003-1330-3254 https://www.mobafire.com/profile/maviyolculukonline12-1122798?profilepage http://hawkee.com/profile/5056616/ https://www.youmagine.com/maviyolculukonline12/designs https://www.myminifactory.com/users/Maviyolculukonline12 http://qooh.me/Maviyolculuko https://linktr.ee/maviyolculukonline12 https://pubhtml5.com/homepage/ivtgi/ https://telegra.ph/Maviyolculukonline12-09-23 https://www.diggerslist.com/650eaa034cb15/about https://allmyfaves.com/Maviyolculukonline12 https://www.metal-archives.com/users/Maviyolculukonline12 https://www.catchafire.org/profiles/2505123/ https://www.fimfiction.net/user/639301/Maviyolculukonline12 https://www.hebergementweb.org/members/maviyolculukonline12.557244/ https://www.sqlservercentral.com/forums/user/maviyolculukonline12 https://www.twitch.tv/maviyolculukonline12/about https://www.roleplaygateway.com/member/Maviyolculukonline12/ https://www.provenexpert.com/maviyolculukonline12/?mode=preview https://www.intensedebate.com/people/Maviyolculukk https://www.indiegogo.com/individuals/35315803 https://visual.ly/users/maviyolculukonline/portfolio https://slides.com/maviyolculukonline12 https://letterboxd.com/Maviyolculukonl/ https://fliphtml5.com/dashboard/public-profile/jpppf https://community.windy.com/user/maviyolculukk https://speakerdeck.com/maviyolculukonline12 https://trello.com/u/maviyolculukonline https://myanimelist.net/profile/Maviyolculukk https://3dwarehouse.sketchup.com/user/7cf754a4-2683-4a00-a629-6b05266c868c/Maviyolculukonline12-M https://www.wattpad.com/user/Maviyolculukonline12 https://www.goodreads.com/user/show/170138048-maviyolculukonline12 https://gravatar.com/maviyolculukonline https://tr.pinterest.com/maviyolculukonline/ https://www.mapleprimes.com/users/Maviyolculukonline12 https://medium.com/@maviyolculukonline https://www.ted.com/profiles/45116335 https://www.infragistics.com/community/members/a2ac6778268e6cc72d99d7ef50004b9f65bbcbbe https://www.metooo.io/u/651138216104c55a8b189d60 https://app.roll20.net/users/12461793/maviyolculukonline12-m https://list.ly/maviyolculukonline/activity https://giphy.com/channel/Maviyolculukonline12 https://www.tumblr.com/maviyolculukonline12 https://www.creativelive.com/student/maviyolculukonline12?via=accounts-freeform_2 https://gab.com/Maviyolculukonline12 https://sketchfab.com/Maviyolculukonline12 https://www.flickr.com/people/199190893@N05/ https://profiles.wordpress.org/maviyolculukonline12/ https://wefunder.com/maviyolculukonline12 https://seedandspark.com/user/maviyolculukonline12-01hb5s51t6xk0p23wrgkgmj27y https://calis.delfi.lv/blogs/lietotajs/315887-maviyolculukonline12/ https://os.mbed.com/users/maviyolculukonline12/ https://notionpress.com/author/929100 https://my.desktopnexus.com/Maviyolculukonline12/ https://guides.co/a/maviyolculukonline12-maviyolculukonline12/ https://www.bahamaslocal.com/userprofile/1/245884/Maviyolculukonline12.html http://molbiol.ru/forums/index.php?showuser=1300115 https://www.credly.com/users/maviyolculukonline12-maviyolculukonline12/badges https://www.facer.io/user/GwGjup0d7n https://ko-fi.com/maviyolculukonline12#paypalModal https://mastodon.online/@Maviyolculukonline12 https://devpost.com/maviyolculukonline?ref_content=user-portfolio&ref_feature=portfolio&ref_medium=global-nav https://www.redbubble.com/people/Maviyolculukk/shop?asc=u https://dzone.com/users/4997525/maviyolculukonline12.html https://hub.docker.com/u/maviyolculukonline12 http://onlineboxing.net/jforum/user/editDone/252629.page https://micro.blog/Maviyolculukonline12 http://www.effecthub.com/user/3728155
Keywords
Lichen, Teloschistaceae, New species, Diversity, Algeria.
Introduction
The Teloschistaceae family is one of the largest families of lichenized fungi, it includes a significant number of genus and species (Lücking et al., 2016). The morphological, anatomical and chemical variation observed for the genus Caloplaca, make a richness with more than 1000 species (Mishra et al., 2020). A new classification of this family has been proposed by Arup et al.,2013, where a total of 39 genera are recognised, of which 31 are newly described. The new genera are: Athallia, Austroplaca, Bryoplaca, Calogaya, Cerothallia, Flavoplaca, Gondwania, Haloplaca, Orientophila, Pachypeltis, Parvoplaca, Rufoplaca, Shackletonia, Scutaria, Sirenophila, Solitaria, Squamulea, Stellaropistangis, Teloschromistora, Teloschromistora. The resurrected genres are Blastenia, Dufourea, Follmannia, Gyalolechia, Leproplaca, Polycauliona, Pyrenodesmia and Xanthocarpia (Arup et al., 2013). The Teloschistaceae are characterised by the presence of parietin as a major by-product which gives a yellowish orange colour to the thallus and apothecia. Polarilocular spores and Teloschistes-type asci are observed. The character of the spores has always been important in determining the species, especially in the genus Caloplaca (Mishra et al., 2020). Teloschistaceae grow in moist, well-lit environments in temperate or Mediterranean climates. They occupy the surfaces and branches of trees of several species. They are found in the corticolous, terricolous and saxicolous types.
Study area
The forest of Doui Thabet has an area of 56.31 km² with a distance of 14 km North-South and 30 km East-West of the province of Saida. It is part of the geographical area of the Tellian highlands. It is limited to the north by the localities of: Sidi Boubkeur and Ouled Khaled, to the west by the locality of Youb, to the east by the town of Saïda and to the south by the locality of Aïn el Hadjar. This forest is composed of four cantons, Sidi Ahmed Zegaï, Oum Touajine, Ras-el-Ma, and El-Ache (Terras, 2011). The bioclimatic Floor is cool semi-arid, with irregular and low rainfall between 300 and 370 mm/year. January and February are the coldest months of the year (3.3°C) while July and August are the hottest (35.5°C) (Baghdadi 2017). The soils of the region are limestone, red, and lithosols (Halitim, 1988). The frequent species in Doui Thabet forest are: Pinus halepensis M., Pistacia lentiscus L., Quercus coccifera L., Ampelodesmos mauritanicus Poir., Rosmarinus tournefortii de noé, Asparagus acutifolius L., Globularia alypum L., Cistus villosus L, Phillyrea angustifolia L., Calycotome spinosa L., Stipa tenacissima L(Terras 2011) (Fig. 1).
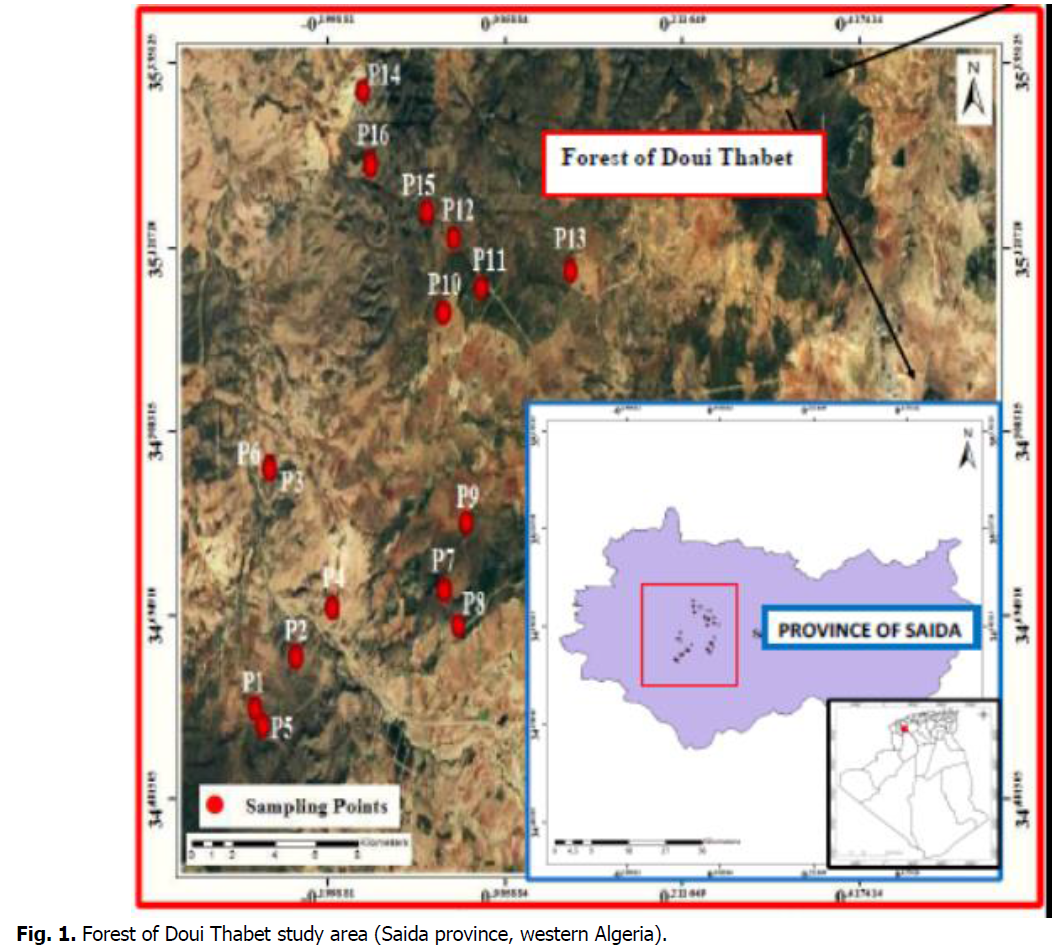
Fig 1: Forest of Doui Thabet study area (Saida province, western Algeria).
Materials and Methods
In the forest of Doui Thabet, we used two sampling methods, the first method for sampling epiphytic lichens. The lichens were counted inside a quadra placed on the tree trunks. We took into account the 4 cardinal points of the tree (Truong, 2013). The information taken on these lichens followed the strict protocols of Wirth, 1997. The second for the sampling of saxicolous and terricolous lichens, we used the partial sampling method. In the field, after having chosen a surface much larger than the minimum area which is (0.5 m2) according to Roux, 1990. The lichens ecology and the nature of the substrate are very important for the lichens identification (calcicole and calcifuge lichens), hydrochloric acid (HCl) is used in the field to identify calcareous and non-calcareous soils and rocks. In the laboratory, specimens were carefully identified by observing their morphology and anatomy using spot tests to determine the compounds in the lichens that are necessary for identification (Kondratyuk et al., 2016). For anatomical studies sections of thalli and apothecia by hand section, spore measurements were performed in K (10% potassium hydroxide) (Holien and Palice, 2018). The spore measurements were estimated in water by calculating: (min-)X-sd-X-X+sd(-max) by the metrology software Piximetre 5.10. We note: (min) and (max) are the extreme values, (X) the arithmetic mean, and (sd) is the corresponding standard deviation. For the identification, determination and nomoclature of lichens, we used several keys and references such as: Les lichens étude biologique et flores illustrée (Ozenda et Clauzade, 1970), The lichens (Wirth, 1995), Guide des lichens de France, lichens des arbres (Van Haluwyn, C., and Asta, J 2017), Guide des lichens de France. Lichens of soils (van Haluwyn, C., Asta, J., and Boissière, J. C. (2014). Guide des lichens de France. Lichens of rocks (Asta, J., van Haluwyn, C., and Boissière, J.C. 2016), The lichens of Italy a second annotated catalogue (Pier Luigi Nimis 2016), Catalogue des lichens et champignons lichénicoles de France métropolitaine (Roux, C. and Coll., E. 2017), as well as the websites Association française de Lichénologie (AFL, 2021), The Information System on Italian Lichens. Version 5.0 (Nimis P.L., 2021), The British Lichen Society (2021) (Fig. 2).
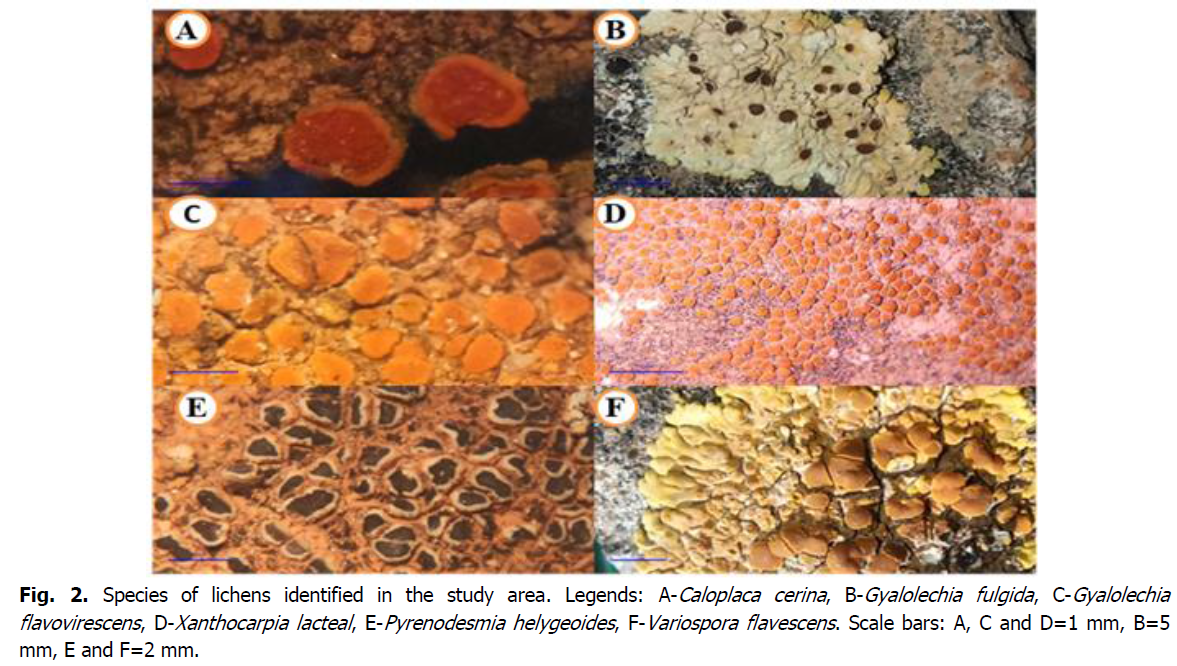
Fig 2: Species of lichens identified in the study area. Legends: A-Caloplaca cerina, B-Gyalolechia fulgida, C-Gyalolechia flavovirescens, D-Xanthocarpia lacteal, E-Pyrenodesmia helygeoides, F-Variospora flavescens. Scale bars: A, C and D=1 mm, B=5 mm, E and F=2 mm.
Results
The determination of the lichenic flora of the teloschistaceae family in the Doui Thabet forest (province of Saïda), revealed twenty-two (22) species of Teloschistaceae divided into 12 genera. The species reported for the first time in Algeria is marked with an asterisk (*). Concerning the classification and synonyms of the taxa, recourse was made to the updated inventory of lichens of Algeria by Ait Hammou et al., (2014), and to Lichenological exploration of Algeria II by Amrani et al. (2018).
List of abbreviations used
Abundance-dominance (AD), Populus sp (P), Pinus halepensis (PHA), Pistacia lentiscus (PL), Quercus coccifera (QC), Tetraclinis articulata (Ta), Crustacean (Cr), Foliaceous (Fo), Corticolous (Co), Terricolous (Tr), Saxicolous (Sa), Siliceous Rock (RS), Calcareous Rock (RC), Calcareous Soil (SC).
*Blastenia xerothermica Vondrák, Arup and I.V. Frolov.
Blastenia is a genus of lichens in the Teloschistaceae family, the origin of Blastenia is dated to the beginning of the Tertiary Age. The genus Blastenia contains 24 species with two subspecies, each species strongly prefers a single substrate type, seventeen (17) species are found on organic substrates, and seven (7) on siliceous rocks, all groups tend to have a Mediterranean distribution, but some epiphytic species have much wider geographical ranges. Only (7) species have vegetative diaspores; they also produce apothecia, but with smaller spores (Vondrák etal., 2020). Blastenia xerothermica is a recently described species; it grows on the branches of trees and shrubs, more rarely on trunks. In the Mediterranean region there is a similar species, Blastenia hungarica. These two species have a different distribution and altitudinal range; note that Blastenia xerothermica is related to the Eu-Mediterranean scrub vegetation (Nimis, 2021).Also it differs by its more developed thallus, its rather visible black pycnidia, and its habitat mainly on deciduous trees (Roux et al., 2017).In Algeria, two species are reported, the first is Blastenia crenularia Gandoger (1883), Flagey (1888, 1895), Egea et al., (1990), Egea and Limona (1991), and the second is Blastenia ferruginea Durieu (1846), Nylander (1854, 1858b, 1878), Flagey (1895, 1896), Paris (1871), Hue (1891), Steiner (1902), Werner (1949), Djellil (1989), Alonso and Egea (2003), Bendaikha (2006), Rebbas et al., (2011), Boutabia et al., (2015), Bendaikha and Hadjadj-Aoul (2016), Boutabia (2016), (Amrani, et al., 2018).
Etymology
This species occupies dry and warm xerothermic habitats.
Description
Crustose, whitish, episubstratic, grey, thin thallus less than 100 μm thick forming patches to 1 cm in diameter. Apothecia biatorine, pale orange or orange-red, sessile, 0.4-0.8 mm in diameter, with a generally flat disc and a smooth, somewhat paler, thin proper margin, thalline margin absent, Proper exciple prosoplectchymatous, orange in outer part, colourless inside, amyloid, epithecium orange-brown, granular 50-70 μm, hymenium colourless, absence of oil droplets 13-35 μm, paraphyses sparingly branched, 1.5-2 μm thick at the in lower part. Apical cells slightly swollen, hypothecium colourless to pale yellowish brown, 38-65 μm, asci Teloschistes-type, octosporate, clavate to cylindrical 44 x 5-12 μm, spores with septum, polarilocular, hyaline, ellipsoid, (8.2-) 8.7- 12.4 (12.9) x (4.3) 4.3-6.1 (-6.2) μm, the septum 4-6 μm thick, photobiont chlorocoid.
Chemical reactions
Thallus R-; apothecium, epithecium K+ red, excipulum and hymenium I+ blue (amyloid).
Similar species
Blastenia hungarica From the lower meso-Mediterranean stage to the supra-Mediterranean stage (Roux and al., 2017)lichen found on branches of acid-barked trees, including oaks, Blastenia ferruginea species apothecia C+ red, excipulum is I-, grows on deciduous trees, equatorial thickening 6-8 µm, Blastenia herbidella thallus consisting of grey isidia.
Collected site
On branches of Tetraclinis articulata, north east exposure, altitude 842m, location 34°55'48''N, 00°01'45''W, canton of El-ache, Doui-Thabet forest, Youb, Saïda province, Algeria, date: on (24/03/2021). Sites: 1, 2, 4, 7, 8 and 9 (Cr, Co, TA, QC, AD1), 10, 11, 12 and 13 (Cr, Co, PL, AD2), 13, 14, 15 and 16 (Cr, Co, QC, AD3).
Ecology
On branches of deciduous trees and shrubs and conifers, more rarely on trunks, mainly at lower altitudes in the Mediterranean scrubland.
Geographical distribution
Blastenia xerothermica is limited to the Mediterranean basin, in Albania, southern France, Greece, Italy, Spain and Turkey (Vondrák etal., 2020).
Caloplaca cerina (Hedw.) Th. Fr. s.lat.
Syn: Blastenia ferruginea var. omora A. Massal, Callopisma cerinum (Hedw.) DeNot, Callopisma cerinum var. effusum A. Massal, Caloplaca cerina f. dispersa H. Olivier, Caloplaca cerina var. nigromarginata (Bagl and Carestia) Jatta, Caloplaca gilva (Vain.) Zahlbr, Caloplaca gilvolutea (Nyl.) Zahlbr, Lecanora gilvolutea Nyl Placodium cerinum (Hedw.) Hepp Placodium gilvum Vain (Nimis, 2022).
Literature: Described by Durieu (1846), Nylander (1854), Paris (1871), Nylander (1878), Gandoger (1883), Flagey (1888, 1895, 1896), Hue (1891), Battandier et al., (1914), Werner (1949), Semadi (1989), Djellil (1989), van Haluwyn and Letrouit-Galinou (1990), van Haluwyn et al.,(1994), Semadi and Tahar (1995), Semadi et al., (1997), Alonso and Egea (2003), Rahali (2003), Ajaj et al.,(2007), Ait Hammou et al., (2011, 2013), Serradj et al., (2013), Slimani et al., (2013), Boutabia et al., (2015), Lograda et al.,(2015), Boutabia (2016), Bendaikha and Hadjadj-Aoul (2016), Ghennam and Abdoun (2017).
Collected site: 1, 2, 4, 7, 8 and 9 (Cr, Co, TA, QC, AD1), 10, 11, 12 and 13 (Cr, Co,PL, AD2), 13, 14, 15 and 16 (Cr, Co, QC, AD1). On smooth, mineral-rich bark, but also on moderately eutrophied bark of other trees, rare in polluted areas.
Caloplaca interna Poelt and Nimis
Description: Crustose, areolate, somewhat thicker epi-substratic thallus, growing on the thallus of often sterile crustose lichens such as Heteroplacidium fusculum and Circinaria sp. Apothecia orange to yellowish-orange, mostly grouped side by side, usually 2-4 apothecia per areole with a rounded to irregular margin, 0.7-0.9 mm in diameter, with a flat orange disc, and a fairly thick orange or reddish orange margin. Thalline margin developed just at base of apothecia, medulla rich in algae, orange epithecium 22.29-34.49 μm K+ red, hymenium colourless, 57-88 μm high I+ blue, paraphyses 2.03-2.5 μm thick at base, becoming progressively thicker towards the top, the apical cells swollen 3.13-4.56 μm wide, colourless hypothecium 84-127 μm. Asci octospores, clavate, Teloschistes-type 11.50-13.90 × 50.43-70.64 μm, thollus somewhat thick 2.15-2.70 μm thick with a blue I+. Spores are polarilocular with septation, colourless, ellipsoid of (10.7) 11-12.9 (13) × (6.1) 6.5-7.3 (7.6) µm, the septum thicker almost1/3 of spore length 3.09-3.87 µm thick.
Chemical reaction: Thallus R-, apothecia, epithecium K+ red, hymenium, asci wall, thollus I+ blue, Photobiont is Chlorococcoides 12.7-17.69 µm in diameter.
Similar species: Flavoplaca oasis (A. Massal.): endolithic thallus, on the thallus of Bagliettoa sp, a calcicolous species growing on hard limestone, the septum of spores up to 7 µm thick.
Collected site: On vertical surfaces of southern slopes on siliceous rocks, develops on the thallus of the often sterile crustacean lichens Heteroplacidium fusculum and Circinaria sp, southern exposure, altitude: 975 m, location: 34°54'56''N, 00°00'30''W, canton of Ras el ma, forest of Doui-Thabet, Youb, province of Saida, date: Algeria on (12/06/2021). Sites: 1, 2, 3, 4 (Cr, Sa, RS, AD2), 13 and 14 (Cr, Sa, RS, AD3), 10, 11 and 12 (Cr, Sa, RS, AD4).
Ecology:On south-facing vertical surfaces on siliceous rocks wetted by water seepage after rain, often found near stands of Peltula (the host being mainly sterile) (Nimis, 2021).
Caloplaca rubelliana (Ach.) Lojka.
Synonym: Callopisma rubellianum (Ach.) A. Massal.
Literature: Described by Flagey (1888, 1895, 1895), Harmand (1913), Werner (1949), Egea et al., (1990), Egea and Llimona (1991).
Collected site: 7, 8 and 9 (Cr, Sa, RS, AD4), 1 and 4 (Cr, Sa, RS, AD3), It is found on steeply sloping surfaces of siliceous rocks in sunny environments.
Flavoplaca oasis (A. Massal.) Arup, Frödén and Søchting f. lithophila auct.
Synonym: Caloplaca holocarpa auct. ital. p.p, Caloplaca lithophila auct. non H. Magn, Caloplaca luteoalba var. saxicola (Hepp) H. Olivier, Flavoplaca oasis A. Massal.) Arup, Frödén and Søchting f. lithophila auct(Nimis, 2022).
Literature: Described by Rahali (2003), Haina et Bendechach (2004).
Collected site:7, 8 and 9 (Cr, Sa, RC, AD2), 12 (Cr, Sa, RC, AD3), 12, 13 and 14 (Cr, Sa, RC, AD1).
Gyalolechia flavovirescens (Wulfen) Søchting, Frödén and Arup
Synonym: Biatora flavofusca (Schrad.) W. Mann, Callopisma aurantiacum var. flavovirescens (Wulfen) A. Massal, Callopisma flavovirescens f. detritum A. Massal, Caloplaca aurantiaca var. inalpina (Ach.) H. Magn, Caloplaca erythrella (Ach.) Kieff, Caloplaca flavorubescens subsp. flavovirescens (Wulfen) Clauzade and Cl. Roux, Caloplaca flavovirescens (Wulfen) Dalla Torre and Sarnth, Caloplaca flavovirescens var. dinae Sbarbaro, Laundonia flavovirescens (Wulfen) S. Y. Kondr, L. Lőkös and J.-S. Hur, Lecanora erythrella (Ach.) Ach, Placodium aurantiacum var. flavovirescens (Wulfen) Hepp, Placodium aurantiacum var. inalpinum (Ach.) H. Magn.
Literature: Described by Durieu (1846), Nylander (1854), Paris (1871), Gandoger (1883), Flagey (1888, 1896), Hue (1891), Steiner (1902), Lapie (1909), Bouly de Lesdain (1911)Faurel et al., (1953c), Egea et al., (1990), Haina et Bendechach (2004).
Collected site: 2, 3, 4, 5, 7, 8 and 9 (Cr, Sa, RC, AD3), 12 (Cr, Sa, RC, AD2), 12, 13 and 14 (Cr, Sa, RC, AD1). A mainly temperate species, it is found on weakly calcareous sandstone and shale, on boulders and walls, at relatively low altitudes.
Gyalolechia fulgens (Sw.) Søchting, Frödén and Arup
Synonym: Caloplaca fulgens (Sw.) Körb, Fulgensia fulgens (Sw.) Elenkin; Fulgensia vulgaris A. Massal. and De Not, Lecanora fulgens (Sw.) Ach, Placodium fulgens (Sw.) DC, Psoroma fulgens (Sw.) A. Massal, Squamaria fulgens (Sw.) Hook, Squamaria fulgens var. decipiens Anzi (Nimis, 2022).
Literature: Described by Montagne (1838), Boivin (1840), Durieu (1846), Nylander (1854, 1858b, 1878), Flagey (1888, 1891, 1896), Hue (1891), Steiner (1902), Maheu (1906), Bouly de Lesdain (1911), Werner (1949), Ajaj et al. (2007), Rebbas et al. (2011).
Collected site: 7, 8 and 9 (Cr, Sa, RC, AD2), 12 (Cr, Sa, RC, AD3), 12, 13 and 14 (Cr, Sa, RC, AD1). It is found on limestone and thin soil layers, often in rock crevices.
Gyalolechia fulgida (Nyl.) Søchting, Frödén and Arup
Synonym: Caloplaca fulgida (Nyl.) J. Steiner, Fulgensia fulgida (Nyl.) Szatala, Lecanora fulgida (Nyl.) Hue (Nimis, 2022).
Literature: Described by Paris (1871), Haina et Bendechach (2004).
Collected site: 2, and 7 (Cr, Sa, RC, AD+). It is mostly found in cracks, more rarely on the ground in dry grasslands.
Kuettlingeria erythrocarpa (Pers.) I.V. Frolov, Vondrák and Arup
Synonym:Blastenia arenaria sensu. Massal, ABlastenia ferruginea var. metabasis A. Massal, Blastenia lallavei (Clemente ex Ach.) A. Massal, Callopisma arenarium auct, Caloplaca arenaria auct. p.p. non (Pers.) Müll. Arg, Caloplaca erythrocarpa (Pers.) Zwackh, Caloplaca erythrocarpa f. diffractoareolata B. de Lesd, Caloplaca lallavei (Clemente ex Ach.) Flagey, Kuettlingeria lallavei (Clemente ex Ach.) Trevis, Placodium lallavei (Clemente ex Ach.) Anzi (Nimis, 2022).
Literature: Described byFlagey (1896), Egea et al., (1990), Haina and Bendechach (2004), Rebas et al., (2011).
Collected site: 3, 5, 9, 10, 11, 12, 14, 15 and 16 (Cr, Sa, RC, AD4), 7 and 8 (Cr, Sa, RC, AD3). It is found on limestone, dolomite, and sandstone, much more rarely on mortar and brick, on horizontal to slightly sloping faces.
Kuettlingeria teicholyta (Ach.) Trevis
Synonym:Blastenia teicholyta (Ach.) Bausch, Blastenia visianica A. Massal, Caloplaca arenaria auct. ital. p.p. non (Pers.) Müll. Arg, Caloplaca erythrocarpa auct. p.p. non (Pers.) Zwackh, Caloplaca teicholyta (Ach.) J. Steiner, Caloplaca visianica (A. Massal.) Jatta, Kuettlingeria visianica (A. Massal.) Trevis (Nimis, 2022).
Literature: Described by Steinheil (1834), Boivin (1840), Durieu (1846), Nylander (1854, 1858b, 1864), Paris (1871), Flagey (1888, 1891, 1896), Hue (1891), Steiner (1895), Bouly de Lesdain (1911), Werner (1949).
Collected site: 9, 10, 11, 12, 14, 15 and 16 (Cr, Sa, RS, AD4), 2, 3, 7 and 8 (Cr, Sa, RS, AD3). Considered an ancient coloniser in warm exposed areas found on calciferous substrates, very rare on pure limestone, commonly found on sandstone and mortar, especially on artificial substrates, walls, roofing tiles, brick walls.
Polycauliona polycarpa (Hoffm.) Frödén, Arup and Søchting
Synonym: Massjukiella polycarpa, Parmelia parietina var. polycarpa (Hoffm.) Fr, Physcia parietina var. pulvinata A. Massal, Xanthoria lychnea var. polycarpa (Hoffm.) Th. Fr, Xanthoria parietina var. polycarpa (Hoffm.) Nyl, Xanthoria polycarpa (Hoffm.) Rieber (Nimis, 2022).
Literature: Described by Flagey (1888, 1895, 1896), Hue (1900), Harmand (1909), Ait Hammou et al., (2008, 2013), Serradj et al., (2013), Boutabia et al., (2015), Lograda et al., (2015), Bendaikha and Hadjadj-Aoul (2016), Khedim et al., (2018).
Collected site: 2 (Fo, Co,PL, AD1). A species found on isolated trees and branches exposed to the sun, on wooden poles, near agricultural and livestock areas, very nitrophilic.
Pyrenodesmia alociza (A. Massal.) Arnold
Synonym: Caloplaca alociza (A. Massal.) Mig, Lecaniella alociza (A. Massal.) Jatta, Lecanora variabilis f. ecrustacea Nyl, Placodium variabi var. ecrustaceum (Nyl.) Nyl, Sporoblastia alocyza (A. Massal.) Trevis (Nimis, 2022).
Literature: Described by Flagey (1888, 1896).
Collected site: 2, 3, 4, 5, 7, 9, 11 and 12 (Cr, Sa, RC, AD4), 13, 14, 15 and 16 (Cr, Sa, RC, AD3). A species found on hard limestone rocks in sunny and exposed areas, it is the most common species of the genus Pyrenodesmia.
Pyrenodesmie helygeoides (Vain.) Arnold
Synonym: Callopisma diphyodes (Nyl.) Bagl. and Carestia sensu Baglietto and Carestia, Caloplaca diphyodes auct. ital. non (Nyl.) Jatta, Caloplaca diphyodes var. helygeoides (Vain.) H. Olivier, Caloplaca diphyodes var. helygeoides (Vain.) H. Olivier; Caloplaca helygeoides (Vain.) Dalla Torre and Sarnth (Nimis, 2022).
Literature: Formerly recognized as a Caloplaca diphyodes,described by Flagey (1896).
Collected site: 1, 2, 3, 7, 8, 9, 11 and 12 (Cr, Sa, RS, AD3), 13, 14, 15 and 16 (Cr, Sa, RS, AD2). Found on siliceous rocks, a non-aquatic species similar to Kuettlingeria diphyodes.
*Pyrenodesmia microstepposa (Frolov, Nadyeina, Khodos. and Vondrák) Hafellner and Türk
The genus Pyrenodesmia is characterised by its crustose, endolithic to epilithic, thick thallus with short marginal lobes. The colour of the thallus and apothecia varies between grey and brown, most lichens of the family Teloschistaceae (Ascomycota) produce yellow, orange, red anthraquinone pigments, however, the genus Pyrenodesmia includes species in which the anthraquinone is absent and replaced by a grey pigment (Sedifolia grey) (Frolovet al., 2021). Vegetative diaspores are present in five species; apothecia are produced by all species, but are less frequent in the soridiate, blastidiate species. This genus is found on inorganic substrates, usually on calcareous rocks (Vondrák et al., 2019).
In Algeria the species reported are Pyrenodesmia agardhiana var. albopruinosa Flagey (1896), Pyrenodesmia agardhiana var. bullosa Flagey (1896), Pyrenodesmia chalybaea (Fr.) A.Massal Flagey (1896), Werner (1949), Rebbas et al., (2011), (Amrani, etal., 2019).
Pyrenodesmia microstepposa
Syn: Caloplaca microstepposaFrolov, Nadyeina, Khodos. and Vondrák (Frolov etal., 2016).
Description: Crustose, epilithic thallus, grey to grey-white, forming small rounded patches up to about 1 cm in diameter or irregular patches up to several cm wide, thallus thickness is (447.4) 501.6-593.3-638.4 (649.2) μm, the algal layer is (35.2) 35.23-49.29-58.3 (65.2) µm thick, the upper cortex is (14.2) 28.1-31.7-41.2 (49.8) µm, and the thickness of the medulla is (355.4) 380.3-445.2-452.1 (461) µm Apothecia, zeorine or rarely biatorine, (0.52) 0.53-0.57-0.65 (0.72) mm in diameter, mature apothecia adnate, rarely immersed or sessile, disc brown to black, thallin margin concolorous to the thallus (53.8)-77-81.5 (96.6) µm in diameter, proper margin brown to black, epruinose disc diameter 564.5-501.8 µm, epithecium usually brown to grey-brown, 33.3-44.7 µm, hymenium colourless 69.4-89.6 μm high, hypothecium colourless, with a central conical extension, 93.4-121.5 µm, paraphyses rarely branched and anastomosed, the upper apical cells usually dead, the lower 1.5-2.5 µm, gradually widening to the upper 3.5-5 µm, asci clavate, octospores of Teloschistes type 60.7-72 × 11.7-14.3 µm, spores eight per asci, colourless, polarilocular, (13.1) 13.11-17.29 (17.3) × (6.3) 7.3-8.5 (8.7) µm, slightly rounded at the end, the septum (0.9) 0.95-1.53-2.4 (2.5) µm wide, cytoplasmic channel in the equatorial thickening is still quite wide at (3.1) 3.14-3.2 (3.4) µm.
Chemical reactions: Thallus and apothecia R-, epithecium K+ brown-violet, hymenium I+ blue, hypothecium I+ blue. Photobiont chlorococcid of (7) 8.45 -9.1-10.1 (10.3) µm.
Similar species: Caloplaca micromontanaFrolov, Wilk and Vondrák. The thallus is generally thinner, less than 150 μm, Apothecia 0.2-0.5 mm, this species is known for Europe and Asia as a mountain species at altitudes between 1700 and 2100 m.
Collected site: On pebbles and small stones in limestone, north-east exposure, altitude: 842 m, location: 34°55'48''N, 00°01'45''W, canton of El-ache, Doui-Thabet forest, Youb, province of Saïda, Algeria on (10/05/2021). Sites: 2, 3, 4, 5, 7, 9, 11 and 12 (Cr, Sa, RC, AD4), 13, 14, 15 and 16 (Cr, Sa, RC, AD3).
Ecology: Pyrenodesmia microstepposa is known from the arid interior and semi-arid regions of Asia and from the dry interior throughout Europe at altitudes up to 1000 meters. This species usually grows on calcareous pebbles and stones, rarely on concrete, often in sunny and sun-exposed environments and in rocky steppes (Frolov et al., 2016).
Rufoplaca arenaria (Pers.) Arup, Søchting and Frödén
Synonym: Blastenia arenaria (Pers.) A. Massal, Blastenia lamprocheila (DC.) Arnold, Caloplaca arenaria (Pers.) Müll. Arg, Caloplaca craspedia (Ach.) Szatala, Caloplaca ferruginascens (Nyl.) H. Olivier, Caloplaca lamprocheila (DC.) Flagey, Lecanora lamprocheila (DC.) Nyl (Nimis, 2022).
Literature: Described by Boivin (1840), Durieu (1846), Nylander (1854), Flagey (1888, 1891, 1896), Steiner (1902), Harmand (1913), Werner (1949), Faurel et al.,(1953b), Esnault and Roux (1987), Djellil (1989), Egea et al., (1990), Rebbas et al., (2011), Lograda et al., (2015).
Collected site: 9 and 8 (Cr, Sa, RS, AD1), 10 (Cr, Sa, RS, AD+). Found on calcified siliceous rocks.
*Usnochroma carphineum (Fr.) Søchting, Arup and Frödén
The genus Usnochroma Søchting Arup and Frödén, is characterised by a crustos, pale yellow thallus without anthraquinones, the etymology of this taxon is by the presence of usnic acid which gives the thallus the yellowish green color. The apothecia are zeorines with a thin proper margin and a rusty orange disc, polarilocular spores with a very long septum. The thallus contains gyrophoric acid and usnic acid, while the apothecia contain parietin. The genus Usnochroma comprises two species: Usnochroma carphineum (Fr.) Søchting, Arup and Frödén et Usnochroma scoriophila(A. Massal.) Sochting, Arup and Froden (Arup et al., 2013).
The genus Usnochroma is reported for the first time in Algeria by the species Usnochroma carphineum in the forest of Doui Thabet.
Synonym: Amphiloma carphineum (Fr.) Bagl, Callopisma carphineum (Fr.) Trevis, Caloplaca carphinea (Fr.) Jatta, Physcia carphinea (Fr.) A. Massal, Squamaria carphinea (Fr.) Boistel (Nimis, 2022).
Description: Thallus crustose, epilithic, placodioid, orbicular, well attached to the substrate, yellowish green to grey-white, 25-30 mm in diameter, the lobes 0.75-1.1 mm wide, long and narrow, fissured areolate, slightly wider at the tips, tightly attached to the substrate, with a black hypothallus. Apothecia zeorine, sessile 0.5-0.8 mm in diameter, with a rusty-red to brown disc 0.43-0.45 mm, with a thin thalline margin of the same colour as the thallus 0.02 mm, and a proper margin lighter than the disc 0.43-0.45 mm. Epithecium orange-brown, 23-47 µm thick, K+ red, hymenium colourless, 60-99 µm high, hypothecium colourless 52-140 µm thick, bounded at the base by an algal layer 37-98 µm thick, paraphyses simple 2.79-3.45 µm in the lower part and sparsely branched on the upper part, the apical cells slightly swollen 3.25-4.28 µm. Asci clavate, octospores of Teloschistes type. Size 67-106 × 12-19 µm. Spores one-septate, ellipsoid, colourless, polarilocular, (10.5) 10.7- 12.3 (12.9) × (6.2) 6.3- 7.2 (7.8) µm, the septum 2.31-3.1 µm. Chlorococcoides photobiont 10.36-10.72 µm in diameter. Chemical reactions: Thallus R-, epithecium K+ purple red, asci and hymenium I+ blue (amiloid).
Collected site: Found on large walls of subvertical siliceous rocks exposed to the sun, southern exposure, altitude: 975 m, location: 34°54'56''N, 00°00'30''W, Canton of Ras el ma, forest of Doui-Thabet, Youb, province of Saïda, Algeria on (12/07/2021) Loc. 6, 7, 8, 9, 10 and 11 (Cr, Sa, RS, AD2).14 and 16 (Cr, Sa, RS, AD1).
Ecology: Found on subvertical, sun-exposed cliffs and basaltic escarpments in warm regions of southern Europe. Found in the thermo- and meso-Mediterranean (AFL, 2021), and subtropical to mild temperate, on hard siliceous rocks in exposed sites (Nimis, 2021).
Similar species: Dimelaena oreina differs by the black colour of the apothecia disc and the uniseptate brown spores (AFL, 2021). Usnochroma scoriophila has an upper cortex thickness of 50-80 µm, imbued with yellow grains, which makes it particularly detachable. The algal layer is more fragmented with protruding parts (Breuss, 1989).
Variospora aurantia (Pers.) Arup, Frödén and Søchting
Synonym: Amphiloma aurantium (Pers.) Müll. Arg, Amphiloma callopisma (Ach.) Körb, Callopisma vulgaris De Not, Caloplaca aurantia (Pers.) Hellb, Caloplaca aurantia var. intermedia Zahlbr, Caloplaca aurantia var. papillata Poelt, Caloplaca callopisma (Ach.) Th. Fr, Gasparrinia aurantia (Pers.) Syd, Gasparrinia callopisma (Ach.) Syd, Klauderuiella aurantia (Pers.) S. Y. Kondr. and J.-S. Hur, Lecanora callopisma Ach, Placodium aurantium (Pers.) Vain, Placodium callopismum (Ach.) Mérat (Nimis, 2022).
Literature: Described by Paris (1871), Flagey (1888, 1891, 1896), Steiner (1902), Bouly de Lesdain (1911), Andreánszky (1934), Werner (1949), Rebbas et al., (2011), Lograda et al., (2015).
Collected site: 1, 3 and13 (Cr, Sa, RC, AD+).
Variospora flavescens (Huds.) Arup, Frödén and Søchting
Synonym: Amphiloma heppianum Müll. Arg, Caloplaca brevilobata (Nyl.) Zahlbr, Caloplaca flavescens (Huds.) J.R. Laundon, Caloplaca heppiana (Müll. Arg.) Zahlbr, Caloplaca sympagea (Ach.) Sandst, Gasparrinia heppiana (Müll. Arg.) Verseghy, Klauderuiella flavescens (Huds.) S. Y. Kondr. and J.-S. Hur; Lecanora heppiana (Müll. Arg.) Hue, Lecanora sympagea Ach, Physcia callopisma var. centroleuca A. Massal, Physcia heppiana (Müll. Arg.) Arnold, Physcia murorum var. centrifuga A. Massal, Physcia murorum var. detrita A. Massal, Placodium callopismum var. brevilobatum (Nyl.) A.L. Sm, Placodium callopismum var. plicatum (Wedd.) Leight, Placodium flavescens (Huds.) A.L. Sm, Placodium heppianum (Müll. Arg.) Flagey (Nimis, 2022).
Literature: Described by Flagey (1888, 1891, 1896), Egea (1988), Egea et al. (1990).
Collected site: 1, 3, 4, 5, 6, 10, 11 12, 13, 14 and15 (Cr, Sa, RC, AD4), 7 and 8 (Cr, Sa, RC, AD2). A common species found on limestone, dolomite, calciferous sandstone, sometimes also on synthetic substrates such as brick, mortar, tile, and on walls.
*Variospora glomerata (Arup) Arup, Søchting and Frödén
The genus Variospora currently comprises twelve species and has recently been separated from Caloplaca s.lat. characterized by a more or less fertile thallus without orange pigments. The species with lobed thallus resemble those of the genus Calogaya or even Rusavskia, but can be separated by the citriform spores or by the equatorial thickening of the spores. The crustacean species can be confused with several other species. By the presence of sublobes or microlobes on the thallus (Arup et al., 2013).
In Algeria five species of the genus variospora are reported, variospora aurantia, variospora dolomiticola, variospora flavescens, variospora hallincola, variospora velana (Amrani, Mark R. D. Seaward, et al., 2019), of which two species are reported for the first time from the Saida province variospora aurantia, variospora flavescens.
Discription: Epilitic crustose thallus consisting of adjacent areoles, yellowish orange, flat and polygonal, thick or slightly convex, angular, some areoles rounded and convex. Apothecia zeorina lecanorine round to irregular in outline deformed by lateral pressure, immersed in areoles 0.4 to 1.5 mm in diameter, clustered so that they can cover much of the thallus flat to slightly convex red-orange disc, a slightly paler, smooth, very thin thallin margin, concolorous to the thallus, well-developed proper margin of the same color of the disc prosoplectenchimatous structure, yellowish-brown epethecium 33-39 μm, colourless hypothecium 33-55um, paraphyses simple articulated rarely branched or anastomosed 1.23-2.17, apical swollen cells 3.13-4.48 μm thick, asci Teloschistes-type, octospores, clavate to cylindrical 56.80 × 7.71-14.64 µm, spores one septum, polarilocular, hyaline, ellipsoid to slightly rhomboid, (10.9) 11.4-13.2 (14.1) × (3.8) 4.9-6.5 (7.3) μm, with thick apical walls, swollen at the septum, which is 4.24-4.97 µm thick. Photobiont chlorococcid green alga 12.14-12.69 μm in diameter.
Chemical reactions: Thallus, apothecia, epithecium C-, K+ purple red. Hymenium, wall of asci, thollus I+ violet blue (amyloid).
Similar species: Xanthocarpia interfulgens apothecies Sessile 0.3-0.5 (-0.6) mm, spores one septum, 14.5-20 (-23) × 5-7 (-8) µm, the septum 1-3 µm. Caloplaca adelphoparasitica apothecia 0.2-0.4 mm diameter, growing on Caloplaca cretensis, Xanthocarpia crenulatella spores narrowly ellipsoid, 13-21 × 4.5-8.5 μm, the septum 1-3 μm thick, parasitic on Verrucaria nigrescens when young (Nimis, 2021). This species is morphologically similar to Caloplaca dolomiticola but differs in spores with thick apical walls, Caloplaca latzelii has biatorial apothecia and an endolithic thallus (Halici et al., 2014).
Collected site: Found on invasive limestone outcrops of two species Rinodinella controversa and Heteroplacidium fusculum, north-western exposure, altitude: 817 m, location: 34°52'55''N, 00°05'17''E, Canton of Sidi Ahmed Zegaï, Doui-Thabet forest, Youb, Saïda province, Algeria on (10/05/2021). Sites: 1, 3, 4, 10, 11 12, 13, 14 and 15 (Cr, Sa, RC, AD4), 5, 6, 7 and 8 (Cr, Sa, RC, AD3).
Ecology: A mainly Mediterranean species found on limestone in circinaria calcarea communities, often invading the thallus of various species of the Pyrenodesmia variabilis group; probably most widespread in southern Italy (Nimis, 2021). This species is described from the Swedish islands of oland and gotland where it seems to be abundant at these localities (Navarro-Rosinés et Hladun, 1992).Saxicolous, calcicolous, on limestone rocks, xerophilous or mesophilous, more or less heliophilous, heminitrophilous. Mesomediterranean, supramediterranean and hill stages (Roux et al., 2017).
Xanthocarpia lactea (A. Massal.) A. Massal
Synonym: Blastenia lactea (A. Massal.) Trevis, Caloplaca lactea (A. Massal.) Zahlbr, Gyalolechia calcicola Galløe nom. Inval, Gyalolechia lactea (A. Massal.) Arnold, Placodium pyraceum var. lacteum (A. Massal.) A.L. Sm (Nimis, 2022).
Literature: Described by Flagey (1896), Bouly (1911), Faurel et al., (1953c), Werner (1949, 1955), Haina and Bendechach (2004).
Collected site: 1, 3, 5, 6, 7, 8, 12, 13 and 14 (Cr, Sa, RC, AD3), 15 and 16 (Cr, Sa, RC, AD2). Found on small limestone pebbles in open habitats on stony ground in dry grasslands.
Xanthocarpia marmorata auct
Synonym: Caloplaca lactea f. fulva (Harm.) Zahlbr, Caloplaca lactea f. laetior J. Steiner, Caloplaca lactea f. rubra (B. de Lesd.) Zahlbr, Caloplaca marmorata auct. non (Bagl.) Jatta, Gyalolechia lactea f. rubra B. de Lesd, Lecanora lactea f. fulva Harm (Nimis, 2022).
Literature: Described by Bouly de Lesdain (1911).
Collected site: 1, 3, 5, 6, 7 and 8 (Cr, Sa, RC, AD2), 12, 13, 14, 15 and 16 (Cr, Sa, RC, AD1). This species is an early coloniser of limestone pebbles in dry grasslands.
Xanthoria parietina (L.) Th.Fr
Synonym: Parmelia parietina (L.) Ach, Parmelia parietina var. vulgaris Schaer, Physcia parietina (L.) De Not, Physcia parietina var. microphylla (Flot.) Körb, Physcia parietina var. platyphylla (Wallr.) Körb. (Nimis. 2022).
Literature: Described by Steinheil (1834), Boivin (1840), Durieu (1846), Nylander (1854, 1858b, 1878), Jourdan (1866, 1867), Semadi and Tahar (1995), Semadi et al. (1997), Tounsi and Benhabyles (2003), Serradj et al. (2013), Slimani et al. (2013), Boutabia et al. (2015), Lograda et al. (2015), Ghennam and Abdoun (2017).
Collected site: 1, 2, 4, 7, 8, 9, 10, 11 and 12 (Fo, Co, Ta, PH, PL, AD4), 13, 14, 15 and 16 (Fo, Co, PH, PL, AD3). Mainly epiphytic found on well-lit trees (Fig. 3-7).
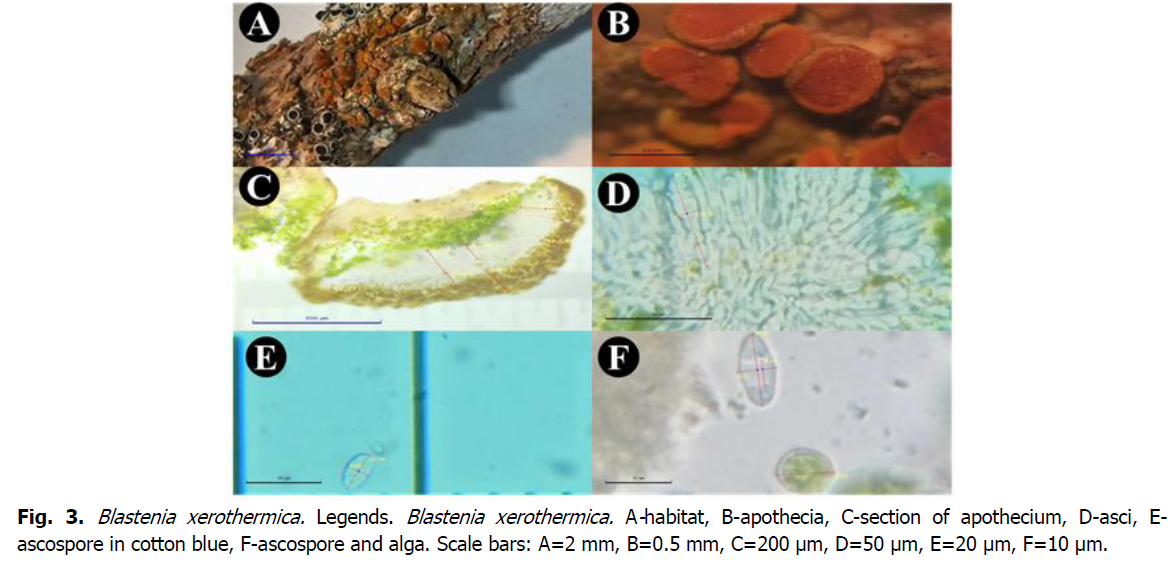
Fig 3: Blastenia xerothermica. Legends. Blastenia xerothermica. A-habitat, B-apothecia, C-section of apothecium, D-asci, E-ascospore in cotton blue, F-ascospore and alga. Scale bars: A=2 mm, B=0.5 mm, C=200 μm, D=50 μm, E=20 μm, F=10 μm.
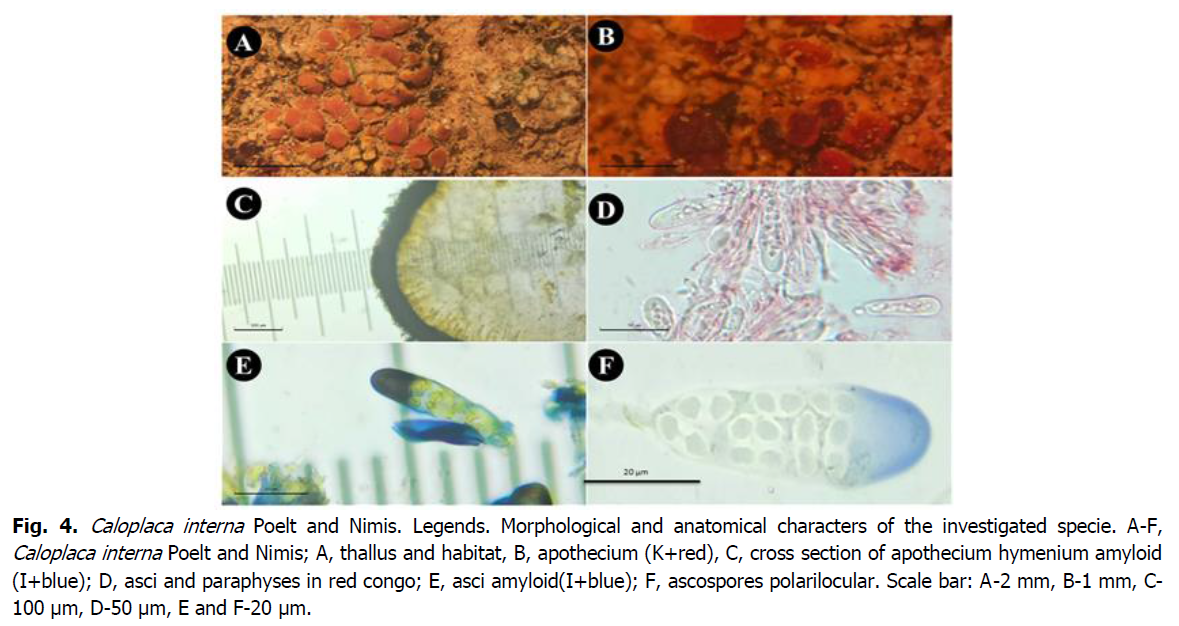
Fig 4: Caloplaca interna Poelt and Nimis. Legends. Morphological and anatomical characters of the investigated specie. A-F, Caloplaca interna Poelt and Nimis; A, thallus and habitat, B, apothecium (K+red), C, cross section of apothecium hymenium amyloid (I+blue); D, asci and paraphyses in red congo; E, asci amyloid(I+blue); F, ascospores polarilocular. Scale bar: A-2 mm, B-1 mm, C-100 μm, D-50 μm, E and F-20 μm.

Fig 5: Pyrenodesmia microstepposa. Legends: Morphological and anatomical characters of the investigated specie. A-H, Pyrenodesmia microstepposa; A, thallus and habitat, B, apothecium, C, cross section of apothecium; D, cross section of thalus; E, Photobiont chlorococcoid; F, Paraphyses in cotton blue; G, ascospores 1-septate, polarilocular, hyaline, ellipsoid; H, asci 8-spored, clavate, Teloschistes-type. Scale bar: A-5 mm, B-0.2 mm, C-200 μm, D-500 μm, E-10 μm, F-50 μm, G-10 μm, H-20 μm.
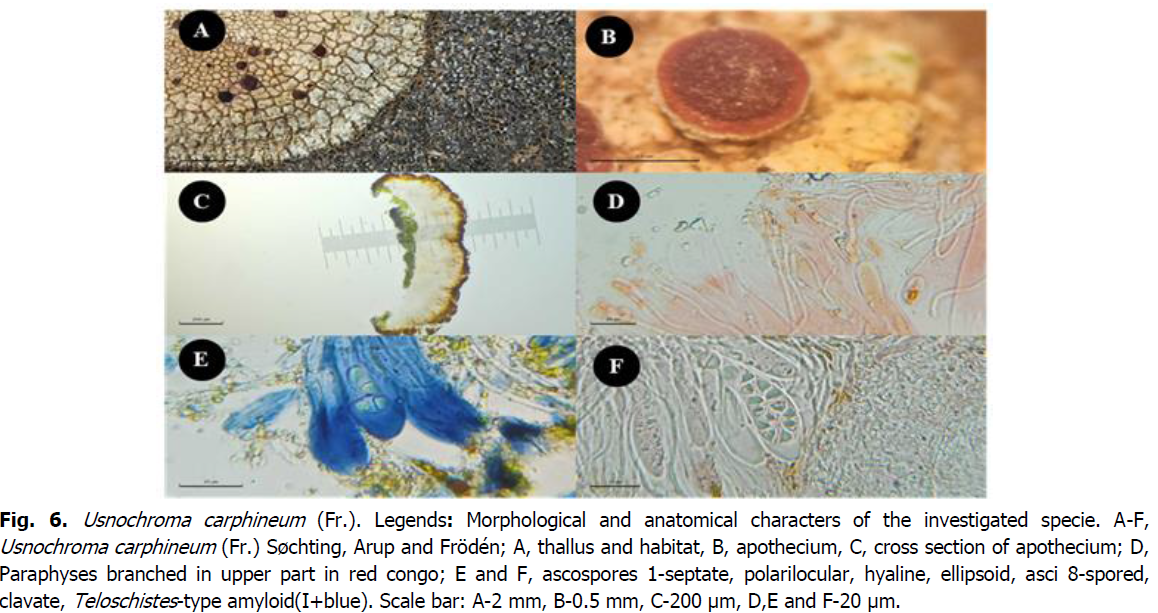
Fig 6: Usnochroma carphineum (Fr.). Legends: Morphological and anatomical characters of the investigated specie. A-F, Usnochroma carphineum (Fr.) Søchting, Arup and Frödén; A, thallus and habitat, B, apothecium, C, cross section of apothecium; D, Paraphyses branched in upper part in red congo; E and F, ascospores 1-septate, polarilocular, hyaline, ellipsoid, asci 8-spored, clavate, Teloschistes-type amyloid(I+blue). Scale bar: A-2 mm, B-0.5 mm, C-200 μm, D,E and F-20 μm.
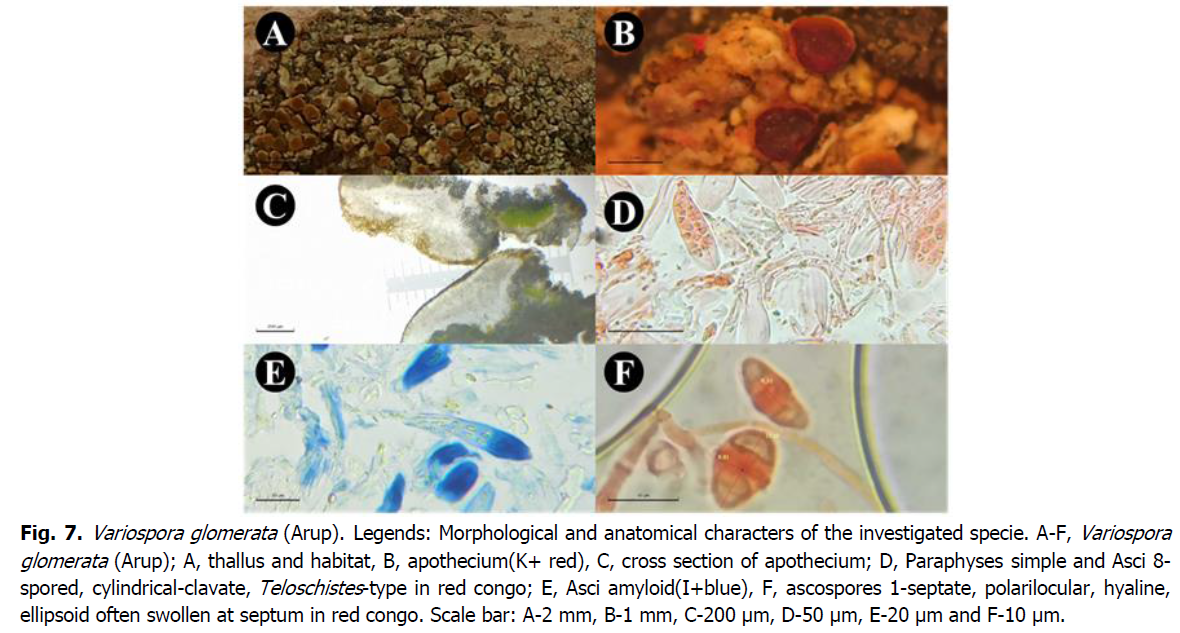
Fig 7: Variospora glomerata (Arup). Legends: Morphological and anatomical characters of the investigated specie. A-F, Variospora glomerata (Arup); A, thallus and habitat, B, apothecium(K+ red), C, cross section of apothecium; D, Paraphyses simple and Asci 8-spored, cylindrical-clavate, Teloschistes-type in red congo; E, Asci amyloid(I+blue), F, ascospores 1-septate, polarilocular, hyaline, ellipsoid often swollen at septum in red congo. Scale bar: A-2 mm, B-1 mm, C-200 μm, D-50 μm, E-20 μm and F-10 μm.
Conclusion
The diversity of Teloschistaceae in the Doui Thabet forest (Saida province, western Algeria) is very abundant on diverse substrates. They are found in this region on terriculous and saxicolous types. During our sampling mission, five new species were recorded for Algeria, as follows: Blastenia xerothermica, Caloplaca intern, Pyrenodesmia microstepposa, Usnochroma carphineum, and Variospora glomerata. The identification also revealed other species of the family Teloschistaceae reported for the first time for this region. They are listed as follows: Caloplaca cerina, Caloplaca rubelliana, Flavoplaca oasis, Gyalolechia flavovirescens, Gyalolechia fulgens, Gyalolechia fulgida, Kuettlingeria erythrocarpa, Kuettlingeria teicholyta, Polycauliona polycarpa, Pyrenodesmia alociza, Pyrenodesmie helygeoides, Rufoplaca arenaria, Variospora aurantia, Variospora flavescens, Xanthocarpia lactea, Xanthocarpia marmorata, Xanthoria parietina. The search for lichenic diversity constitutes a very important richness in medical potential. These species are very fragile and very resistant to the most hostile environments; their presence constitutes a good marker of global climate change.
References
AFL (Association française de Lichénologie). (2021).http://www.afl-lichenologie.fr/.
Ahmed, A.S., Yahia, N., Okkacha, H. (2020). Ecological characterization and evaluation of the floristic potential of the forest of Doui Thabet (Saida-Western Algeria) in the context of the restoration’. 26:266-278.
Amrani, S., Seaward, M.R., Sipman, H.J., Feuerer, T. (2018). Lichenological exploration of Algeria II: checklist of lichenized, lichenicolous and allied fungi. Herzogia, 31:817-892.
Arup, U., Søchting, U., Frödén, P. (2013). A new taxonomy of the family Teloschistaceae. Nordic Journal of Botany, 31:16-83.
Asta, J., Van Haluwyn, C., Bertrand, M. (2016). Guide des lichens de France: Lichens des roches. Éditions Belin.
Baghdadi, K. (2017). Inventaire de la végétation de Djebel El-Ach dans la wilaya de Saïda en perspective d’un aménagement sylvo-cynégétique. Master’s Thesis, University of Saïda.
BLS (British Lichen Society). (2021). https://www.britishlichensociety.org.uk/.
Braun-Blanquet, J. (1964). Pflanzensoziologie. Aufl Wien.
Breuss, O. (1989). Zur Unterscheidungvon Caloplaca carphinea und C. scoriophila (Lichenes, Teloschistaceae). Linzerbiology Beitr, 21:583-590.
Frolov, I., Vondrák, J., Fernández-Mendoza, F., Wilk, K., Khodosovtsev, A., Halıcı, M.G. (2016). Three new, seemingly-cryptic species in the lichen genus Caloplaca (Teloschistaceae) distinguished in two-phase phenotype evaluation. In Annales Botanici Fennici, Finnish Zoological and Botanical Publishing Board, 53:243-262.
Frolov, I., Vondrák, J., Košnar, J., Arup, U. (2021). Phylogenetic relationships within Pyrenodesmia sensu lato and the role of pigments in its taxonomic interpretation. Journal of Systematics and Evolution, 59:454-474.
Halitim, A. (1988). Sols des régions arides d’Algérie. O.P.U, Alger, pp:1-75.
Holien, H., Palice, Z. (2018). Lecidea subhumida Vain, a pine wood specialist new to Scandinavia’. 30:59-64.
Kondratyuk, S.Y. (2016). Fauriea, a new genus of the lecanoroid caloplacoid lichens (teloschistaceae, lichen-forming ascomycetes)’. Acta Botanica Hungarica, 59:303-318.
Lücking, R., Hodkinson, B.P., Leavitt, S.D. (2016). The 2016 classification of lichenized fungi in the Ascomycota and Basidiomycota–approaching one thousand genera. The Bryologist, 119:361-416.
Mishra, G.K. (2020). Current taxonomy of the lichen family teloschistaceae from India with descriptions of new species’. Acta Botanica Hungarica, 62:309-391.
Mohamed, A.H., Miara, M.D., Rebbas, K., Slimani, A., Ravera, S., Hamerelain, A.S. (2014). Mise à jour de l’inventaire des lichens d’Algérie. Revue Ecologie-Environment, 10:75-103.
Nimis, P.L. (2016). ITALIC-The information system on italian lichens. Version 5.0. University of Trieste, Department of Biology.
Nimis, P.L. (2016). The lichens of Italy. A second annotated catalogue. EUT Edizioni Università di Trieste.
Ozenda, P., Clauzade, G. (1970). Les Lichens, étude biologique et flore illustrée. Paris-Vie: éd. Masson et Cie.
Roux, C. (1990). Echantillonnage de la végétation lichénique et approche critique des méthodes de relevés. Cryptogamie Bryology Lichénology, 11:95-108.
Roux, C., Coll, E. (2017). Atalogue des lichens et champignons lichénicoles de France métropolitaine. 2 Edition Revue et Augmentée.
Talbi, O., Benabdeli, K., Benhanifia, K., Hadouche, D. (2017). Cartographie des zones de risque de feux de forêt dans la commune de Doui Thabet, Saïda, Algérie. International Journal of Environmental Studies, 75:543-552.
Terras, M. (2011). Typologie cartographie des stations forestières et modélisations des peuplements forestiers. Cas des massifs forestiers de la wilaya de Saida (Algérie). Tlemcen: Doctorat thesis, University of Tlemcen.
Truo, N.G.C. (2013). Les lichens de la ville de Neuchâtel-Inventaire et étude de bio-indication. Bulletin Social Neuchâtel Science Nature, 133:21-48.
Van Haluwyn, C., Asta, J. (2017). Guide des lichens de France, lichens des arbres mise à jour 2017.
Van Haluwyn, C., Asta, J., Boissière, J.C. (2014). Guide des lichens de France. Lichens des sols: Lichens des sols. Belin éditeur.
Vondrak, J., Frolov, I., Davydov, E.A., Yakovchenko, L., Malíček, J., Svoboda, S., Kubásek, J. (2019). The lichen family Teloschistaceae in the Altai-Sayan region (Central Asia). Phytotaxa, 396:1-66.
Vondrák, J., Frolov, I., Košnar, J., Arup, U., Veselská, T., Halıcı, G., Søchting, U. (2020). Substrate switches, phenotypic innovations and allopatric speciation formed taxonomic diversity within the lichen genus Blastenia. Journal of Systematics and Evolution, 58:295-330.
Wirth, V. (1995), The lichens. 1:1-165.
Author Info
R. Mouedden1*, A. Ayache1, A.H. Benchohra2, F. Toumi2 and F.Z. Bendimred12Department of Environmental Sciences, Faculty of Natural and Life Sciences, Eco-development Laboratory for Spaces, University of Djillali liabes, Sidi Bel Abbes 22000, Algeria
Citation: Mouedden, R., Ayache, A., Benchohra, A.H., Toumi, F., Bendimred, F.Z. (2022). Five new species of teloschistaceae lichens from Algeria. Ukrainian Journal of Ecology. 12:74-86.
Received: 21-Feb-2022, Manuscript No. UJE-22-55033; Accepted: 16-Mar-2022, Pre QC No. P-55033; Editor assigned: 23-Feb-2022, Pre QC No. P-55033; Reviewed: 01-Mar-2022, QC No. Q-55033; Revised: 07-Mar-2022, Manuscript No. R-55033; Published: 23-Mar-2022, DOI: 10.15421/2022_356
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
