Research - (2021) Volume 0, Issue 0
Impact of climatic variability on the duration of phenological phases, productivity, and essential oil quality of Dracocephalum moldavica L. in agroclimatic zones of Polissya and Forest-Steppe in Ukraine
L.A. Kotyuk1*, I.V. Ivashchenko1, O.А. Коrablova2 and D.B. Rakhmetov2Abstract
The impact of climatic variability on the phenological phases of Dracocephalum moldavica change was studied under introduction in two agroclimatic zones of Ukraine - Poland and Forest-Steppe. Field, introductory, acclimatization, biomorphological, biochemical, and statistical methods were used in the research. It was found that the general duration of a life cycle of D. moldavica under conditions of Polissya ranged from 137 to 143 days and in Forest-Steppe from 122 to 153 days. The plants of D. moldavica under the conditions of Polissya during the vital cycle required 1138.2° of effective temperatures and in Forest-Steppe 1384.6 ° (average in 2011-2013). Insufficient precipitation and soil moisture supply impacted germination rates and plant development in the period of germination and vegetative growth. On the contrary, high summer temperatures and the lack of precipitation resulted in the reduction period of flowering and fruiting. The productivity of the aboveground mass was 2.5 ± 0.3 kg/m2 in Polissya and 2.3 ± 0.2 kg/m2 in Forest-Steppe. We recorded that the essential oil content was 0.715 ± 0.133% in Polissya and 0.652 ± 0.142% in Forest-Steppe. The content of the main components in the essential oil of D. moldavica was higher in the Polissya zone than in Forest-Steppe. Therefore, the climatic variability and modern natural conditions of the Polissya and Forest-Steppe zones corresponded fully to the biological needs of D. moldavica for moisture and a specific thermal regime during the vegetative period. The introduction of D. moldavica plants into industry culture in these regions is promising.
Keywords
Dracocephalum moldavica, essential oil, introduction, phenological phases, productivity, the sum of effective temperatures.
Introduction
Climate variability has become a global issue as it can negatively affect different systems and sectors and threaten human well-being (Somboonsuke et al., 2018; Fitter & Fitter, 2002). Recently, climate variability had a noticeable impact on many areas of life. There are growing concerns about possible climate variability and its further impact on agriculture. Crop production is inherently sensitive to climate variability. Temperature is a significant determinant of the rate of plant development and, under climate change. A warmer temperature has shortened the development stages of crops and will most probably reduce the total yield (Apiratikorn et al., 2021). The impacts of climate change on plant productivity will be significantly influenced by how climate affects the rate of plant development and the duration of their growth. Earlier crop flowering and maturity have been observed and documented in recent decades, and these are often associated with warmer temperatures (Craufurd & Wheeler, 2009; Tao et al., 2006; Hu et al., 2005; Williams et al., 2004). The timing of flowering and seed ripening occurred earlier at high temperatures, but caused a reduction in the duration of the vegetative stage of plant growth and a decrease in fruit yield (Fitter & Fitter, 2002). 78% of all observations in European countries were reported to show earlier flowering, with an advance in phenological events of 2.5 days per decade on average (Estrella et al., 2007; Menzel et al., 2006).
Changes in the duration of the growing season concern agricultural, phenological, economic, epidemiological, and bioclimatological reasons (Vega et al., 2020). Analysis of phenological changes in vegetation is essential to assess the response and adaptation of agroecosystems to climate change (Bandoc et al., 2017; Wolski & Kwiatkowski, 2006). The climate in Ukraine is changing faster than in the world. Since 1991, every decade in Ukraine has been warmer than the previous. The average annual temperature in Ukraine in 1961–1990 was + 7.8° C and at the end of the last decade (2010–2019), it has already reached the level of +9.6°C. Due to the increase in average air temperatures, Ukraine received additional heat resources - the sum of active temperatures above +10°C (Adamenko, 2020; Ackermann, 2020). This is an excellent opportunity to expand the area under aromatic and medicinal plants that love heat, which were previously grown mainly in the southern regions of Ukraine. Due to its unique biological and biochemical properties, D. moldavica is one of the most common species among Lamiaceae family plants in a culture of the European-Asian continent. The habitat for D. moldavica plants is Turkey and Iran. In nature, the plant occurs in Middle Asia (Turkmenistan), West and East Siberia, in the Far East, Mongolia, China, and North America. In Europe, the plant was introduced into culture as an essential oil plant (Aimovi et al., 2019). In Ukraine (Korablоva & Rakhmetov, 2012; Takhtadjian, 2009).
Dracocephalum moldavica L. (Moldavian dragonhead) is an annual grassy plant, 30–80 cm high, with a thin taproot, an orthotropic tetrahedral branched stem, oppositely placed leaves, cluster inflorescences with white, blue, or purple flowers (Kotyuk, 2013 ; Korablova, 2011). D. moldavica is grown industrially in the southern regions (Steppe zone) for the perfumery, pharmaceutical and food industries (Zeng et al., 2018; Jiang et al., 2014). Now the cultivation of aromatic crops is gradually shifting to the north (Kotyuk, 2016; Ovechko et al., 2001). A broad application of D. moldavica plants is explained by their high content of vitamins, proteins, lipids, total sugars, tannins, macro- and microelements in the above ground mass. The value of D. moldavica plants is the synthesis of essential oil used in the food industry, perfumery, and cosmetology (Shahrajabian et al., 2020; Kotyuk, 2013a; Rakhmetov et al., 2004). Essential oil from D. moldavica plants contains the components used as anti-inflammatory and sedative remedies to fight off a cold, headache, neuralgia, rheumatism, and joint pain to cure tachycardia, hypertension, and insomnia (Shahrajabian et al., 2020; Carović-Stanko et al., 2016). Component geraniol–an acyclic monoterpene alcohol mixture of the two cis-trans isomers appropriately named geraniol (trans) and nerol (cis). It is emitted from the flowers of many species, and it is present in the vegetative tissues of many herbs and often coexists with geranial and neral, which are the oxidation products of geraniol (Aćimović et al., 2019).
The raw material of D. moldavica is used in industry to make vermouth, absinth, stewed fruit, aromatization of tea and vinegar; in cooking as a seasoning for chicken and fish dishes, soups, sauces, vegetable side dishes, and salad (Alael et al., 2013; Kotyuk, 2013). It is well known that the periodicity of vital cycles and adaptive properties of introducers depend on abiotic conditions of a natural-climatic zone. A temperature threshold of phenological phases, showing biorhythms dependence on the temperature regime, is an essential quantitative indicator of the dynamics of seasonal plant development; therefore, it is used as a comparative criterion in the analysis studies, connected with the introducer adaptation. A sum of active temperatures indicates heat supply in a vegetative period (Rakhmetov, 2011). It has been established that D. moldavica plants are xerophilous; they have leaves with a thick epidermis that tend to fold along a central vein at heat (Autko et al., 2003). In case of lack of moisture and light, plant productivity abruptly decreases, vegetation extends, and seed and essential oil yield decreases. Thus, a significant factor of regular vital activity of the plant is moisture supply, which is determined by indicators of relative air humidity and precipitation amount (Baykova et al., 2002). The lack of sufficient moisture can cause a stressful situation for plants, so artificial watering of D. moldavica plants (Alael et al., 2013) can be used.
The climatic conditions associated with temperature affect the sowing time, size, and quality of the crop. Therefore, improving the understanding of changes in important temperature indicators has great social and economic importance. Given the limited amount of information on adaptive properties of D. moldavica plants when introduced into culture, the purpose of the research is to identify the effect of abiotic factors on the seasonal rhythms of plant development and the possibilities of industrial cultivation of aromatic plants in the conditions of the Polissya and Forest-Steppe zone of Ukraine.
Materials and Methods
The subject of the research is the plants of Dracocephalum moldavica L. under conditions of Botanical garden of Polissya National University (BG PNU, Zhytomyr), situated in the Polissya zone of Ukraine (latitude N 50°15'07'', longitude E 28°41'53''), and M. M. Gryshko National Botanical Garden of National Academy of Science of Ukraine (NBG NASU, Kyiv), situated in the Forest-Steppe zone (latitude N 50°24'45'', longitude E 30°33'44''). The seed material from a collection of aromatic plants of the cultural flora department of the NBG NASU was used. The plants were cultivated on collection plots of BG PNU and NBG during the 2011–2013 years. The experiments were laid down on open sunny plots. Seeds were sown during late April–early May on a depth of 1.5cm, using a 45 × 30 cm scheme. Plant maintenance during all the years of the research was the same. The soils of experimental plots of BG PNU are dark gray podzolized. Humus content is 2.97 ± 0.08%; Рh-salt humus horizon – 6.2 ± 0.05; P2O5 content – 458.0 ± 4.77 mg/kg; K2O – 84.0 ± 3.86 mg/kg; N – 90.5 ± 3.73 mg/kg of the soil. An experimental plot of NBG has gray forest podzolized soils. Humus content is 3.26 ± 0.07%; Рh-salt humus horizon – 6.7 ± 0.05; P2O5 content – 373 ± 4.87 mg/kg; K2O – 66 ± 2.86 mg/kg; N – 98±2.67 mg/kg of the soil.
The effect of temperature, precipitation, and relative air humidity on the development phase of Dracocephalum moldavica plants, yield, and quality of essential oils was studied. Data from the Ukrainian Hydrometeorological Center were used. We made calculations as to the sum of active and effective temperatures of a vital cycle of Moldavian dragon's head in general and by separate age periods. The average daily values of air temperature were used in the research. The sum of active temperatures was calculated with a formula: Σtаkt=t*п, where: t-active average daily temperature for a period, °С; п–a number of days in a period. The sum of effective temperatures for this period was calculated as Σtеf=(t–В) *п, where: В–biological minimum, 10°С (Polovyi, 2001).
The biological minimum of temperatures for Dracocephalum moldavica is +10°С; below these levels, the development of plants is stopped. The data received was processed statistically using Microsoft Excel 10. Features of development, phenology, life cycles, and aspects of plant introduction were studied according to the methods Beydeman (1974) and Rakhmetov (2011), productivity–Dospekhov (1986), and quality of essential oil–Chernogorod (2002).
Results and Discussion
Plants of D. moldavica go through four age periods and 12 age states under the conditions of introduction. When classifying age states, criteria that characterize mainly qualitative plant features are used (Korablova & Rakhmetov, 2012; Rakhmetov et al., 2004). Seed (se) is a latent period from seed ripening to seed germination. The sprout (p) is a period from the moment the embryo or its part emerges from a seed wall to the appearance of the first true leaves of D. moldavica. Seedling (pl) is a period from the appearance of the first true leaves to cotyledon necrosis. Juvenile state (j) is a period from the onset of cotyledon death to the beginning of the development of lateral shoots of D. moldavica. During this period, plants do not have the features and properties that are characteristic of mature plants. The immaturity state (im) is a period from the beginning of the development of lateral shoots to the appearance of the features of a mature plant. The virginal state (v) is a period from the time the characteristic structures of mature plants dominate to the beginning of the development of generative shoots. Young generative state (g1) is a period from the appearance of generative organs to complete formation of the typical structures of mature plants. In this period, active processes of growth and formation of shoots and a root system take place. The middle age generative state (g2) is characterized by the highest degree of development of a root and shoot system of D. moldavica and the highest seed productivity. The processes of new formations and necrosis are balanced.
The population of D. moldavica plants was estimated using indicators of vegetative and generative recovery, actual seed productivity, and laboratory and field seed germination. Depending on the climatic conditions, the seedlings have started appearing 7-20 days after sowing. D. moldavica plants developed very slowly –from 20 to 30 days –from seedlings to forming the third pair of leaves. The fourth and subsequent pairs of leaves formed in an interval of 4 to 7 days. After that, the development was much faster. D. moldavica plants are characterized by an aboveground type of germination; cotyledons come out of the soil without a seed wall. A typical xeromorphic characteristic of D. moldavica plants is mucus that forms on the seeds when they contact water; this helps retain moisture in a seedling zone and ensures fast germination. In this period, blue flower seedlings have distinct anthocyanin coloring on leaves and stems. Seedlings do not form a rosette, which is why epicotyl is well seen, and it continues intercalary growth for some time after the formation of the first pair of true leaves.
A growth cone in a seedling phase is a thickened tubercle with some first pair of leaves. Leaf primordia are formed on a growth cone during the first pair of true leaves; later, they become the basis for successive leaves. Abscission of the cotyledons and the beginning of the formation of lateral shoots were observed in the formation of five to six pairs of true leaves, that is, the transition of the plants from a juvenile to an immature state. More intensive seedling growth was recorded after abscission of the cotyledons (Kotyuk, 2013b). During the formation of immature and virginal species, one could observe the growth of a stem, and the shoots of second-order (lateral) plants began to develop the characteristics of a manure species. The change in leaf form from oval egg-shaped to oblong egg-shaped is observed in juvenile and immature age states of D. moldavica plants. The formation of leaves is over before flowering; however, the leaf blades continue to grow to reach their maximum assimilating surface in the period of mass flowering (Fig. 1). The reproductive period of D. moldavica plants begins with the formation of flower buds. The appearance of the first buds coincides with the period of intensive growth of a plant, from the end of June to late July. The phase of bud formation continues until the opening of the first flower. Although growth activity somewhat decreases in this period, it lasts until the end of a plant flowering phase.

Figure 1: Dracocephalum moldavica during young generative state (g1), middle-age generative state (g2).
Racemose inflorescences of D. moldavica consist of close rings with six flowers on short pedicles that have bracts with spinous barbs. The axillary leaves get an elongated form with sharp serrated edges. Flowering begins with the main shoot. Usually, flowering of flower lasts for five days, but at mass flowering, it somewhat decreases. A flowering phase of D. moldavica plants is extended in time; it can last up to 50% of a vital cycle and ends in mid-September.
A large time gap between the first and last flower formation is typical for these plants. Inflorescences have a reciprocal type of bloom characterized by the opening of the lowest flower first and the acropetally development of the following flowers. Therefore, flowers can be in the stage of fruit formation on the same shoot in its lower part, whereas they only develop buds and anthers in its upper part.
A peculiar feature of D. moldavica plants is that lateral shoots begin the differentiation of generative organs almost two weeks later than the main shoot, but they go through all stages a bit faster and reach them at the end of the vegetation.
Drying of leaves and stems was recorded after fertilization. The drying of the plants begins in mass flowering when the seeds ripen in a lower part of the central florescence. First, the color of the leaves changes, they become yellow, brown, and gradually they abscise from the lower part, and then the stem withers.
The completion of vegetation is characterized by the total withering of the above and below ground organs. D. moldavica dies in mid-September in the Polissya zone of Ukraine and in early October in the Forest-Steppe zone. 2011 was an exception when D. moldavica plants finished their vegetation in August.
Parshina (2009), Grjaznov & Totskaya (2019), and Totskaya et al. (2013) reported that the duration of the vegetation period of D. moldavica plants in the Almaty and Moscow regions was 140 days and 150 days, respectively. Ovechko et al. (2001) reported that the vegetation period of D. moldavica plants in the Kherson region was 120 to 130 days on average; in the Forest-Steppe zone of Ukraine, it was 103 to 157 days and in the west Podillya 140 to 150 days (Shanaida et al., 2008). In the conditions of the Stavropol Territory, the period from full emergence to the beginning of the flowering of the D. moldavica plant was 105–115 days (Chumakova & Chumakov, 2018), in Yakutia, it was 96–106 days (Egorova, 2017), in Belarus 92–105 days (Savich & Tychina, 2017), in the conditions of the forest-Steppe zone of Ukraine this period was 53–77 days (Shanaida et al., 2008).
During the whole research period, the general duration of a vital cycle of D. moldavica under the conditions of PBG was as follows: in 2011–140 days, 2012-147 days, 2013-143 days, on average it was 140 days; Under the conditions of NBG it was 122 days, 129 days and 153 days, respectively, on average 135 days, which agrees well with the data of the researchers mentioned above (Table 1). Analysis of air moisture indicators in the introduction regions shows their dynamic changes toward increase from April to September - during a vegetation period. For example, the average monthly indicators of air moisture during vegetation periods increased from 56.9% (germination) to 78.0% (necrosis). In the years of the research, the average moisture indicators changed by 5–10% in months.
| Development phase | Sum of effective temperatures,°с | Sum of active temperatures,°с | Relative air humidity, % | Precipitation, mm | Phase duration, days | Sum of effective temperatures,°с | Sum of active temperatures, °с | Relative air humidity, % | Precipitation,mm | Phase duration, days |
|---|---|---|---|---|---|---|---|---|---|---|
| PBG | NBG | |||||||||
| 2011 | ||||||||||
| Seedling | 38.5 | 148.5 | 70.2 | 31.5 | 15 | 214.9 | 638.7 | 62.6 | 27.5 | 28 |
| Vegetative growth | 318.5 | 658.5 | 60.2 | 26.6 | 35 | 175.1 | 325.1 | 46.7 | 2.0 | 15 |
| Budding | 126.8 | 276.8 | 74.7 | 100.1 | 15 | 200.4 | 661.8 | 61.8 | 30.3 | 17 |
| Flowering | 248.7 | 538.7 | 77.0 | 191.9 | 29 | 314.2 | 614.2 | 79.5 | 235.0 | 30 |
| Fruiting | 174.8 | 514.8 | 72.1 | 61.1 | 37 | 223.3 | 443.3 | 75.7 | 68.0 | 22 |
| Necrosis | 23.0 | 73.0 | 74.3 | 12.0 | 9 | 139.0 | 239.0 | 42.0 | 4.1 | 10 |
| 2012 | ||||||||||
| Seedling | 115.5 | 245.5 | 63.4 | 13.0 | 12 | 171.3 | 381.3 | 72.2 | 68.0 | 21 |
| Vegetative growth | 244.2 | 574.2 | 69.3 | 96.4 | 34 | 165.2 | 365.2 | 71.3 | 80.7 | 19 |
| Budding | 182.4 | 362.4 | 68.8 | 23.7 | 18 | 454.9 | 824.9 | 66.3 | 37.8 | 16 |
| Flowering | 373.8 | 683.8 | 69.4 | 59.2 | 31 | 465.8 | 865.8 | 69.4 | 126.6 | 38 |
| Fruiting | 294.7 | 604.7 | 73.3 | 81.9 | 31 | 158.0 | 408.0 | 71.19 | 12,5 | 24 |
| Necrosis | 68.5 | 178.5 | 73.3 | 81.9 | 11 | 119.0 | 225.2 | 79.5 | 23.5 | 11 |
| 2013 | ||||||||||
| Seedling | 134.1 | 314.1 | 56.9 | 0.4 | 18 | 245,3 | 679.3 | 64.7 | 46.8 | 27 |
| Vegetative growth | 312.5 | 682.5 | 72.1 | 47.8 | 37 | 299.0 | 559.0 | 66.7 | 46.3 | 26 |
| Budding | 267.2 | 517.2 | 72.1 | 31.0 | 25 | 200.0 | 360.0 | 68.1 | 48.4 | 16 |
| Flowering | 324.4 | 664.4 | 73.4 | 37.4 | 34 | 434.3 | 834.3 | 66.4 | 29.6 | 40 |
| Fruiting | 129.2 | 339.2 | 75.5 | 81.5 | 21 | 163.1 | 521.1 | 84.0 | 210.8 | 36 |
| Necrosis | 38.0 | 118.0 | 78.0 | 3.4 | 8 | 11.1 | 78.5 | 85.4 | 53.9 | 8 |
| Average for 3 years | ||||||||||
| Seedling | 96.0 | 236.0 | 63.5 | 15.0 | 15 | 278.7 | 566,4 | 66.5 | 47.4 | 25.3 |
| Vegetative growth | 291.7 | 638.4 | 67.2 | 56.9 | 35.3 | 213.1 | 416.4 | 61.6 | 43.0 | 20.0 |
| Budding | 192.1 | 385.5 | 71.9 | 51.6 | 19.3 | 285.1 | 615.6 | 65.4 | 38.8 | 16.3 |
| Flowering | 315.6 | 629.0 | 73.3 | 96.2 | 31.3 | 404.8 | 771.4 | 71.8 | 130.4 | 36.0 |
| Fruiting | 199.6 | 486.2 | 73.6 | 74.8 | 29.7 | 180.9 | 457.5 | 77.0 | 97.1 | 27.3 |
| Necrosis | 43.2 | 123.1 | 75.2 | 32.4 | 9.3 | 89.7 | 180.9 | 69.0 | 27.2 | 9.7 |
Table 1. Effect of climatic factors on the duration of development phases of D. moldavica plants under introduction in PBG and NBG (average for 2011–2013)
Correlation between the duration of development phases of the plants and some abiotic factors was established (Table 2). For example, in the condition of PBG on the average in the years of the research, the highest correlation was between a phase duration and a sum of effective temperatures (r=0.93), precipitation amount had a lower effect (r=0.76).
| PBG | NBG | |||||
| Indexes | Sum of effective temperatures,°С | Relative air humidity, % | Precipitation, mm | Sum of effective temperatures, °С | Relative air humidity, % | Precipitation, mm |
|---|---|---|---|---|---|---|
| Relative air humidity, % | 0.02 | 1.00 | -0.02 | 1.00 | ||
| Precipitation, mm | 0.82 | 0.51 | 1.00 | 0.61 | 0.68 | 1.00 |
| Phase duration, days | 0.93 | -0.06 | 0.76 | 0.77 | 0.45 | 0.91 |
Table 2. Correlation matrix dependence of the duration of development phases of the plants Dracocephalum moldavica for the climatic factors (r).
In the forest-Steppe zone of Ukraine (NBG), the duration of the vital cycles of D. moldavica plants from germination to harvesting raw material for industrial processing and seed harvesting did not differ much from that of the Polissya zone (PBG) (Table 3). On the contrary, in NBG, the amount of precipitation had a greater effect (r = 0.91) and the temperature indicators had a lower effect (r=0.77). Relative air humidity had the lowest effect in both zones, particularly in Polissya (PBG).
| Zone | Comparative duration of the period from germination to | ||
|---|---|---|---|
| Collection of raw materials for technical processing | seed collection | the end of the growing season | |
| Central Polissya | 100.9 ± 3.2 | 130.6 ± 4.4 | 139.9 ± 4.7 |
| Right-Bank Forest-Steppe of Ukraine | 97.6 ± 4.1 | 124.9 ± 5.5 | 134.6 ± 3.9 |
Table 3. Duration of a vital cycle of plants Dracocephalum moldavica depending on the zone of introduction, harvesting terms, and application spheres (days ± SD).
The sum of active temperatures required for the vegetation period of D. moldavica plants in the introduction conditions is present in Figs 2 and 3. According to Ovechko et al. (2001), for the normal development of D. moldavica in the Steppe zone in the south of Ukraine, active temperatures of 2600 to 3500° are necessary. According to Chumakova & Chumakov (2018), the sum of temperatures during the growing season of D. moldavica plants in Stavropol was 2400-2900°, and the relative humidity ranged from 37 to 79%. In Forest-Steppe in Western Siberia, the sum of active temperatures required for the development of D. moldavica was 2262-3153°, and effective temperatures 941-1673°, (Sarlaeva et al., 2016).
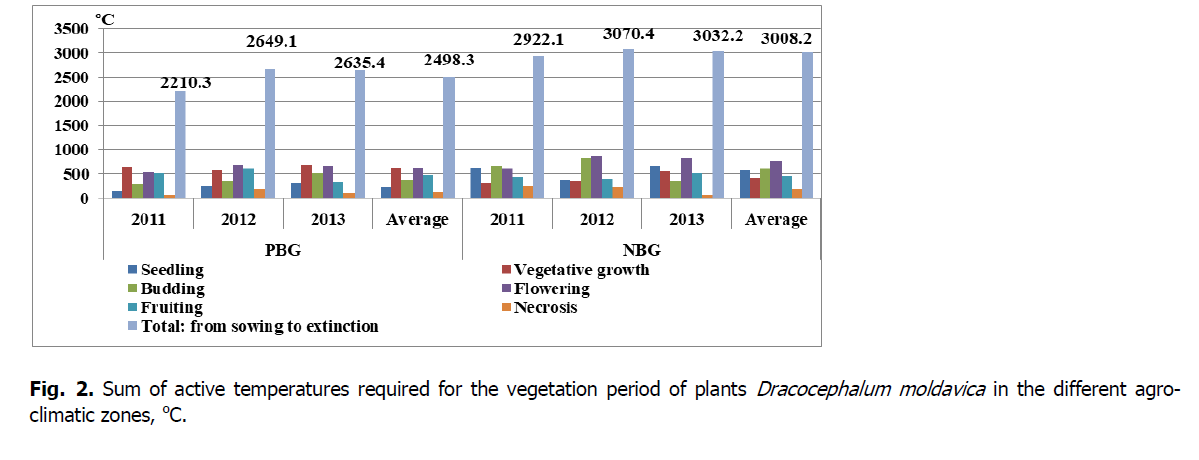
Figure 2: Sum of active temperatures required for the vegetation period of plants Dracocephalum moldavica in the different agro-climatic zones, оС.
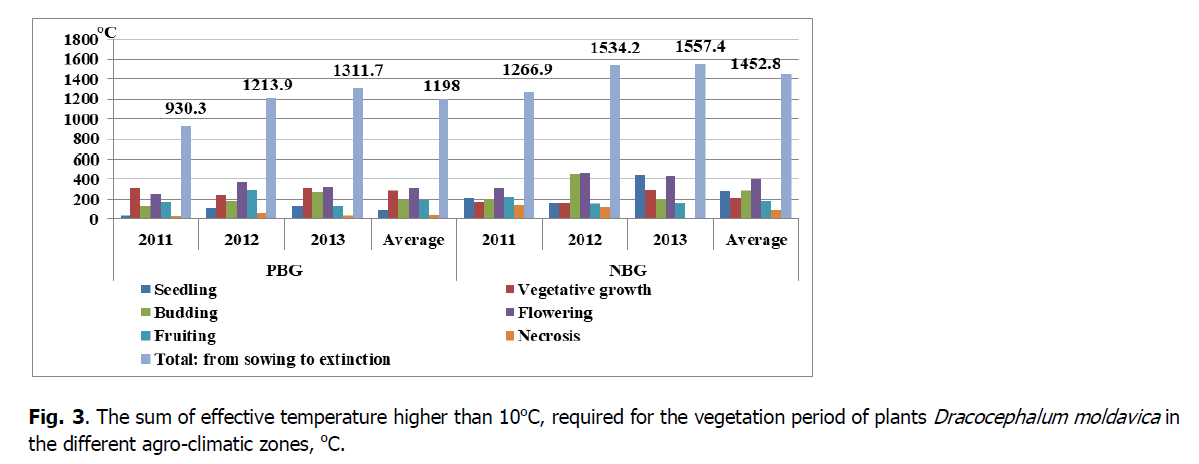
Figure 3: The sum of effective temperature higher than 10оС, required for the vegetation period of plants Dracocephalum moldavica in the different agro-climatic zones, оС.
The duration of a fruiting phase of D. moldavica in the introduction in PBG ranged from 21 days (2013) to 37 days (2011) due to the total lack of precipitation in August 2013. Under the conditions of introduction in NBG, considering a large amount of precipitation and moderate temperature, the longest fruiting phase was recorded in 2013 (36 days). Thus, it was found that a sum of active temperatures for a D. moldavica plant development cycle under the conditions of PBG from sowing to death was 2498.3° on average; the minimum sum of temperatures was 2210.3° (2011), the maximum sum of temperatures was 2649.1° (2012).
In the conditions of NBG for a development cycle of D. moldavica plants from sowing to death, the sum of active temperatures was 3008.2° on average; the minimum sum of temperatures was 2922.1° (2011), the maximum sum of temperatures was 3070.4° (2012).
In PBG, during a vital cycle, D. moldavica plants used on average the sum of effective temperatures of 1138.2° (from 930.3 to 1279.1°) and under the conditions of NBG -1384.6° (from 1266.9 to 1534.2°, respectively).
It is established that D. moldavica plants grown in new conditions have a high yield of 8 aboveground mass and good quality of essential oil (Table 4, Fig. 4).
| Indexes | Polyssia | Forest-Steppe |
|---|---|---|
| Plant height, cm | 71.6 ± 9.3 | 73.5 ± 5.1 |
| Seed yield, g/m2 | 70 ± 3.2 | 45 ± 4.1 |
| Productivity of aboveground mass, kg/m2 | 2.5 ± 0.3 | 2.3 ± 0.2 |
| The content of essential oil, % of the absolute dry weight | 0.715 ± 0.133 | 0.652 ± 0.142 |
Table 4. Comparative analysis of biometric parameters and productivity of D. moldavica plants in Polissya and Forest-Steppe of Ukraine (average data ± SD) for the 2011–2013 year.
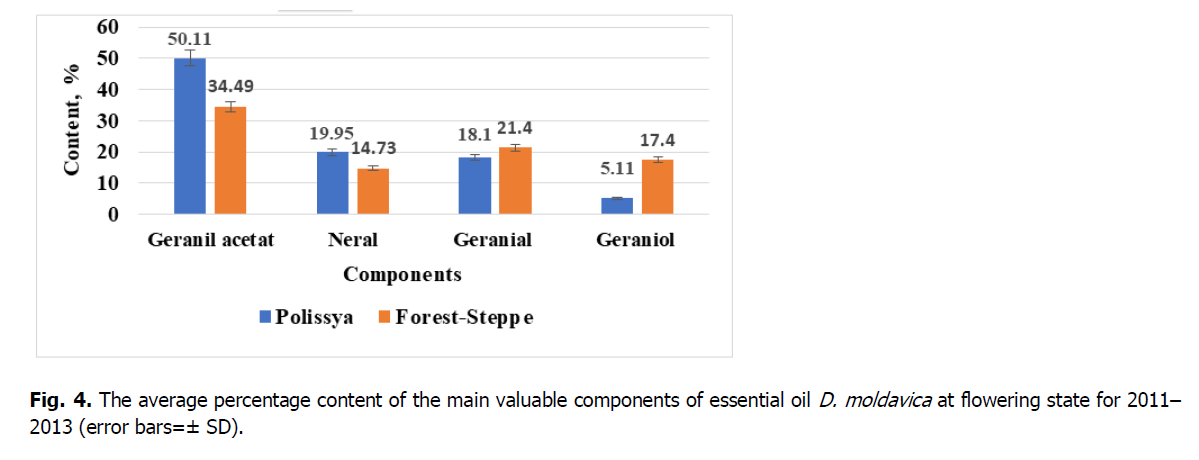
Figure 4: The average percentage content of the main valuable components of essential oil D. moldavica at flowering state for 2011–2013 (error bars=± SD).
It was found that under the conditions of Polissya D. moldavica plants, they give a significantly higher seed yield, while other indicators differ insignificantly.
With an increase in geraniol during the development of inflorescences in plants, geranyl acetate in the oil decreases accordingly. This indicates the interconversion of geranyl acetate into geraniol. This monoterpene alcohol is a widely used fragrance material. A survey of consumer products revealed that it is present in 76% of the deodorants investigated on the European market, included in 41% of household products and 33% of cosmetic formulations based on natural ingredients (Chen et al., 2010; Rastogi et al., 2001). One of the essential oil of the valuable components of D. moldavica essential oil is citral–a mixture of isomers geranial and neral with a predominance of geranial. The citral content in the essential oil of Polissya is 38.05% and in the essential oil of Forest-Steppe 36.13%. Citral is used as a flavoring in the food industry, an antiseptic, anti-inflammatory agent, and a raw vitamin A material.
Conclusion
In this way, the strong influence of the temperature factor on the life cycle of D. moldavica was revealed, duration which in recent years differed little in Polissya and Forest-Steppe zones (140 days and 135 days, respectively). In both zones, relative air humidity had a much smaller impact on the plant's development duration. Insufficient precipitation and moisture supply in the spring affected seedling emergence and plant development rates during the phases of germination and vegetative growth. High summer temperatures and the lack of precipitation caused the reduction of the duration phases of flowering and fruiting. The frost-free period is enough for a vital cycle of D. moldavica plants under Polissya and Forest-Steppe zone conditions, which have considerable agro-ecological importance for Ukraine. Biological needs of plants in moisture amount and a sum of effective temperatures during a vegetative period correspond to natural climatic conditions in these two zones. The plants go through all ontogenesis phases, including fruiting, which proves successful introduction and confirms their high ecologically plasticity and ability to adapt to the new climatic conditions. The yield of grass and quality of essential oil of D. moldavica plants confirms good perspectives to cultivate them in these zones for the national economy's perfumery, medical and food branches. Our results could be used in adapting the other types of crops to new climatic conditions.
References
Aćimović, M., Sikora, V., Brdar-Jokanović, M., Kiprovski, B., Popović, V., Koren, A., Puvača, N. (2019). Dracocephalum moldovica: cultivation, chemical composition and biological activity. Journal of Engineering and Technology Management. 2:153-167.
Ackermann, A. (2020). Climate change threatens the Ukrainian breadbasket.
Adamenko, T. (2020). Kyiv region becomes a steppe zone.
Alael, S., Melicyan, A., Kobraee, S., Mahna, N. (2013). Effect of different soil moisture levels on morphological and physiological characteristics of Dracocephalum moldavica. Agricultural Communications, 1:23-26.
Apiratikorn, S., Sdoodee, S., Lerslerwong, L., Rongsaw, S. (2012). The impact of climatic variability on phenological change, yield and fruit quality of mangosteen in phatthalung province, Southern Thailand Kasetsart J, 46:1-9.
Autko, А.А., Rupasova, G.А., Autko, А.А., Kuhareva, L.V., Suhorska-Trotylo, K. (2003). Bioecological features of growing aromatic medicinal plants. Мinsk, Тоnpik, Belarus.
Bandoc, G., Prăvălie, R., Patriche, C., Dragomir, E., Tomescu, M. (2017). Response of phenological events to climate warming in the southern and south-eastern regions of Romania. Stochastic Environmental Research and Risk Assessment. 32:1113-1129.
Baykova, E.V., Korolyuk, Е.А., Тkachev, А.V. (2002). Component composition of essential oils of some species of the genus Salvia L. grown in Novosibirsk (Russia). Chemistry of Plant Raw Materials. 1:37-42.
Beydeman, I.N. (1974). Methods of studying of phenology of plants and plant communities. Novosibirsk, Nauka. (in Russian).
Carović-Stanko, K., Petek, M., Grdiša, M., Pintar, J., Bedeković, D., Herak Ćustić, M., Satovic, Z. (2016). Medicinal plants of the family Lamiaceae as functional foods-a review. Czech Journal Food Science, 34:377-390.
Chen, W., Viljoen A.M. (2010). Geraniol-A review of a commercially important fragrance material. South African Journal of Botany. 76:643-651.
Chernogorod, L.B., Rabotyagov, V.D., Vinogradov B.A. (2002). Dynamics of composition of essential oil Achillea collina Beck. (ex Reich) in ontogenesis. Plants Introduction. 3:127–132.
Chumakova, V.V., Chumakov, V.F. (2018). Agrobiological and phytochemical characteristics of the Moldavian snakehead variety 'Egoist'. Agricultural Magazine, 2:2-32.
Craufurd P.Q., Wheeler T.R. (2009). Climate change and the flowering time of annual crops. Journal Experimental Botany, 60:2529-2539.
Dospekhov, B.A. (1986). Field experiment technique (with the basics of statistical processing of research results). Rev and Add Moscow, Agropromizdat.
Egorova, P.S. (2017). To the introduction of Dracocephalum moldavica L. (Moldavian snakehead) in the Yakutsk Botanical Garden. Krasnoyarsk SAU Bulletin, Russia, 5:117-122.
Estrella, N., Sparks, T., Menzel, A. (2007). Trends and temperature response in the phenology of crops in Germany. Global Change Biology, 13:1737-1747.
Fitter, A.H., Fitter, R.S.R. (2002). Rapid changes in flowering time in British plants. Science. 296:1689-1691.
Grjaznov, M.Yu., Totskaya, S.A. (2019). Dracocephalum moldavica L. new variety of breeding of VILAR. Vegetable crops of Russia. 3:65-67.
Hu, Q., Weiss, A., Feng, S., Baenziger, P.S. (2005). Earlier winter wheat heading dates and warmer spring in the U.S. Great Plains, Agricultural and Forest Meteorology, 135:284-290.
Jiang, J., Yuan, X., Wang. T., Chen, H., Zhao, H., Yan, X., Wang, Z., Sun. X., Zheng, Q. (2014).
Antioxidative and cardioprotective effects of total flavonoids extracted from Dracocephalum moldavica L. against acute ischemia/reperfusion-induced myocardial injury in isolated rat heart. Cardiovasc Toxicology 14:74-82.
Korablova, O.A. (2011). Spices and seasonings. Kyiv, Univest Media.
Korablova, O.A., Rakhmetov, D.B. (2012). Useful plants in Ukraine: from introduction to use. Kyiv, Phytosocial Center.
Kotyuk, L.A. (2013a). Content of ascorbic acid and carotene in raw aromatic plants of the family Lamiaceae Lindl. Studia Biologica. 7:83-90.
Kotyuk, L.A. (2013b). Morphological peculiarities of Dracocephalum moldavica L. with respect to its introduction in the Botanical Garden of ZhNAEU. Modern Phytomorphology, 4:293-297.
Kotyuk, L.A. (2016). Antimicrobial activity of oil-bearing plants Lamiaceae Lindl. towards Escherichia coli. Biological Bulletin of Bogdan Chmelnitskiy Melitopol State Pedagogical University, 6:216-236.
Menzel, A., Jakobi, G., Ahas, R., Scheifinger, H., Estrella, N. (2003). Variations of the climatological growing season (1951–2000) in Germany compared with other countries. International Journal of Climate 23:793-812.
Ovechko, S.V., Svidenko, L.V., Rabotyagov, V.D. (2001). Biology of growth and development Dracocephalum moldavica L. in conditions of Steppe zone on the south of Ukraine. Plant Introduction, 11:71-73.
Parshina, G. (2009). Features of development of Dracocephalum moldavica L. during introduction in Almaty region. Newsletter of the Kiev University. Series: Introductions and conservation of growing growth. 22:13-14.
Polovyi, A.M., Bozhko, L.Yu., Sitov, V.M., Yarmolskaya, O.E. (2001). Workshop on agricultural meteorology. Odessa, TPP.
Rakhmetov, D.B., Stadnichuk, N.О., Korablova, O.A., Smylianec, N.М., Skrypka, О.L. (2004). New fodder, spice and vegetable introducers in the Forest-Steppe and Polyssia of Ukraine. Kyiv, Phytosocial Center, Ukraine.
Rakhmetov, D.B. (2011). Theoretical and practical aspects of plant introduction in Ukraine. Kyiv, Agr. Media Group.
Rastogi, S.C., Heydorn, S., Johansen, J.D., Basketter, D.A. (2001). Fragrance chemicals in domestic and occupational products. Contact Dermatitis, 45:221-225.
Sarlaeva, I.Ya., Baikova, E.V., Vasilieva, O.Yu. (2016). Heat supply of annual ornamental plants in the Forest-Steppe zone of Western Siberia. Advances in Modern Science and Education, 12:188-197.
Savich, I.M., Tychina I.N. (2017). Features of the development of Moldavian snakehead species (Dracocephalum moldavica L.) in the Central Botanical Garden of the National Academy of Sciences of Belarus. In: Proceedings of the International scientific conference dedicated to the 85th anniversary of the Central Botanical Garden of the National Academy of Sciences of Belarus. Minsk, Madisont, Belarus, 1:241-243.
Shahrajabian, M.H, Sun, W, Soleymani, A, Cheng, Q. (2020). Traditional herbal medicines to overcome stress, anxiety and improve mental health in outbreaks of human coronaviruses. Phytotherapy Research. 35:1-11.
Shanaida, M.I., Pasemkiv, Yu.A., Korablova, O.A., Rakhmetov, D.B. (2008). Results of introductory and phytochemical studies of species of the genus Dracocephalum L. (D. grandiflorum L. and D. moldavica L.) under conditions of the Northern Forest-Steppe and the Western Podillya zones of Ukraine. Plant Introduction, 2:65-71.
Somboonsuke, B., Phitthayaphinant, P., Sdoodee, S., Kongmanee, C. (2018). Farmers perceptions of impacts of climate variability on agriculture and adaptation strategies in Songkhla Lake basin. Kasetsart Journal of Social Sciences, 39:277-283.
Takhtajan, A. (2009). Flowering plants, 2nd ed. Springer Science & Business Media B.V. (in Russian). Tao, F., Yokozawa, M., Xu, Y., Hayashi, Y., Zhang Z. (2006). Climate changes and trends in phenology and yields of field crops in China, 1981–2000. Agricultural and Forest Meteorology. 138:82–92.
Totskaya, S.A., Korotkikh, I.N., Khazieva, F.M. (2013). Post-harvest ripening of seeds of the Moldavian snakehead (Dracocephalum moldavica L.). Biological, Medical and 14 Pharmaceutical Chemistry Issues, 5:25-27.
Vega, A.J., Rohli, R.V., Wright, E. (2020). Changes in growing season in the Northeastern United States (U.S). Physical Geography, 41:343-364.
Wolski, T., Kwiatkowski, S. (2006). Biology of growth and development of moldavian dragonhead (Dracocephalum moldavica L.)-aromatic and medicinal plant. Postępy Fitoterapii, 7:2-10.
Williams, T., Abberton, M. (2004). Earlier flowering between 1962 and 2002 in agricultural 21 varieties of white clover. Oecologia. 138:122-126.
Zeng, C., Jiang, W., Yang, X., He, C., Wang, W., Xing, J. (2018). Pretreatment with total flavonoid extract from Dracocephalum moldavica L. attenuates ischemia reperfusion-induced apoptosis. Scientific Reports, 8-17491.
Author Info
L.A. Kotyuk1*, I.V. Ivashchenko1, O.А. Коrablova2 and D.B. Rakhmetov22M.M. Gryshko National Botanical Garden of the NAS of Ukraine, Timiryazevska str., 1, Kyiv, 01014, Ukraine
Citation: Kotyuk, L.A., Ivashchenko, I.V., ??rablova, O.?., Rakhmetov, D.B. (2021). Impact of climatic variability on the duration of phenological phases, productivity, and essential oil quality of Dracocephalum moldavica L. in agroclimatic zones of Polissya and Forest-Steppe in Ukraine. Ukrainian Journal of Ecology, 11 (7), 39-47.
Received: 29-Jul-2021 Accepted: 06-Sep-2021 Published: 27-Sep-2021
Copyright: This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
